Nucleus accumbens: Difference between revisions
→Function: renaming Neurotransmitters |
→Research: breaking this up into 2 sections named Function and Clinical significance. Renamed "Deep brain stimulation" to Depression |
||
| Line 77: | Line 77: | ||
'''Serotonin (5-HT):''' Overall, [[Serotonin|5-HT]] synapses are more abundant and have a greater number of synaptic contacts in the NAc shell than in the core. They are also larger and thicker, and contain more large dense core vesicles than their counterparts in the core. |
'''Serotonin (5-HT):''' Overall, [[Serotonin|5-HT]] synapses are more abundant and have a greater number of synaptic contacts in the NAc shell than in the core. They are also larger and thicker, and contain more large dense core vesicles than their counterparts in the core. |
||
== |
==Function== |
||
| ⚫ | |||
| ⚫ | Although the nucleus accumbens has traditionally been studied for its role in addiction, it plays an equal role in processing many rewards such as food and sex. The nucleus accumbens is selectively activated during the perception of pleasant, emotionally arousing pictures and during mental imagery of pleasant, emotional scenes.<ref name=Costa>{{cite journal |author=Costa, VD, Lang, PJ, Sabatinelli, D, Bradley MM, and Versace, F |title=Emotional imagery: Assessing pleasure and arousal in the brain's reward circuitry |journal=Human Brain Mapping |volume=31 |pages=1446–1457 |year=2010 |doi=10.1002/hbm.20948 |pmid=20127869 |issue=9}}</ref><ref name=Sabatinelli>{{cite journal |author=Sabatinelli, D, Lang, PJ, Bradley, MM, Costa, VD, and Versace, F |title=Pleasure rather than salience activates human nucleus accumbens and medial prefrontal cortex |journal=Journal of Neurophysiology |volume=98 |pages=1374–1379 |year=2007 |doi=10.1152/jn.00230.2007 |pmid=17596422 |issue=9}}</ref> A 2005 study found that it is involved in the regulation of emotions induced by [[music]],<ref name=Menon>Menon, Vinod & Levitin, Daniel J. (2005) ''The rewards of music listening: Response and physiological connectivity of themesolimbic system." ''NeuroImage'' 28(1), pp. 175-184</ref> perhaps consequent to its role in mediating dopamine release. The nucleus accumbens plays a role in [[Cognitive neuroscience of music|rhythmic timing]] and is considered to be of central importance to the limbic-motor interface (Mogensen).{{Citation needed|date=April 2014}} |
||
| ⚫ | In the 1950s, [[James Olds]] and Peter Milner implanted electrodes into the septal area of the rat and found that the rat chose to press a lever which stimulated it. It continued to prefer this even over stopping to eat or drink. This suggests that the area is the "pleasure center" of the brain and is involved in reinforcement learning.<ref>{{cite journal |author=Olds J, Milner P |title=Positive reinforcement produced by electrical stimulation of septal area and other regions of rat brain |journal=J Comp Physiol Psychol |volume=47 |issue=6 |pages=419–27 |year=1954 |pmid=13233369 |doi=10.1037/h0058775}} [http://www.wadsworth.com/psychology_d/templates/student_resources/0155060678_rathus/ps/ps02.html article]</ref> In rats, stimulation of the [[ventral tegmental area]] causes the release of dopamine in the nucleus accumbens much in the same way as addictive drugs and natural reinforcers, such as water or food, initiate the release of dopamine in the nucleus accumbens.<ref>Nakahara, D., Ozaki, N., Miura, Y., Miura, H., et al. Increased dopamine and serotonin metabolism in rat nucleus accumbens produced by intracranial sel-stimulation of medial forebrain bundle as measured by in vivo microdialysis. ''Brain Research'''''', 1989 495: 178-181.</ref> The same results have been seen in human subjects in functional imaging studies. For example, increased dopamine concentration is seen in the extracellular fluid of the nucleus accumbens when subjects believed they were being given money{{Citation needed|date=April 2014}}, and increased activation (i.e., increased fMRI BOLD signal-change) was observed among heterosexual males viewing pictures of attractive women.<ref>Aharon, L., Etcoff, N., Ariely, D., CHabris, C. F., et al. Beautiful faces have variable reward value: fMRI and behavioral evidence. ''Neuron'' 2001, ''32'', 357-551.</ref> |
||
| ⚫ | |||
| ⚫ | An fMRI study conducted in 2005 found that when mother rats were in the presence of their pups the regions of the brain involved in reinforcement, including the nucleus accumbens, were highly active.<ref>Ferris, C.F., Kulkarni, P., Sullivan, J.M., Harder, J.A., et al. Pup sucking is more rewarding than cocaine: Evidence from functional magnetic resonance imaging and three-dimensional computational analysis. ''Journal of Neuroscience'''''', 2005 25: 149-156.</ref> Levels of dopamine increase in the nucleus accumbens during maternal behavior, while lesions in this area upset maternal behavior.<ref>Numan, M. Motivational systems and the neural circuitry of maternal behavior in the rat. ''Developmental Psychobiology'''''', 2007 49: 12-21.</ref> When human mothers are presented pictures of their children, fMRIs show an increased brain activity in the nucleus accumbens and other reinforcing brain regions and a decrease in activity in areas of the brain involved with negative emotions.{{citation needed|date=June 2013}} |
||
==Clinical significance== |
|||
===Addiction=== |
===Addiction=== |
||
| Line 87: | Line 97: | ||
{{:FOSB|Table title=Summary of addiction-related plasticity}} |
{{:FOSB|Table title=Summary of addiction-related plasticity}} |
||
| ⚫ | |||
| ⚫ | Although the nucleus accumbens has traditionally been studied for its role in addiction, it plays an equal role in processing many rewards such as food and sex. The nucleus accumbens is selectively activated during the perception of pleasant, emotionally arousing pictures and during mental imagery of pleasant, emotional scenes.<ref name=Costa>{{cite journal |author=Costa, VD, Lang, PJ, Sabatinelli, D, Bradley MM, and Versace, F |title=Emotional imagery: Assessing pleasure and arousal in the brain's reward circuitry |journal=Human Brain Mapping |volume=31 |pages=1446–1457 |year=2010 |doi=10.1002/hbm.20948 |pmid=20127869 |issue=9}}</ref><ref name=Sabatinelli>{{cite journal |author=Sabatinelli, D, Lang, PJ, Bradley, MM, Costa, VD, and Versace, F |title=Pleasure rather than salience activates human nucleus accumbens and medial prefrontal cortex |journal=Journal of Neurophysiology |volume=98 |pages=1374–1379 |year=2007 |doi=10.1152/jn.00230.2007 |pmid=17596422 |issue=9}}</ref> A 2005 study found that it is involved in the regulation of emotions induced by [[music]],<ref name=Menon>Menon, Vinod & Levitin, Daniel J. (2005) ''The rewards of music listening: Response and physiological connectivity of themesolimbic system." ''NeuroImage'' 28(1), pp. 175-184</ref> perhaps consequent to its role in mediating dopamine release. The nucleus accumbens plays a role in [[Cognitive neuroscience of music|rhythmic timing]] and is considered to be of central importance to the limbic-motor interface (Mogensen).{{Citation needed|date=April 2014}} |
||
| ⚫ | In the 1950s, [[James Olds]] and Peter Milner implanted electrodes into the septal area of the rat and found that the rat chose to press a lever which stimulated it. It continued to prefer this even over stopping to eat or drink. This suggests that the area is the "pleasure center" of the brain and is involved in reinforcement learning.<ref>{{cite journal |author=Olds J, Milner P |title=Positive reinforcement produced by electrical stimulation of septal area and other regions of rat brain |journal=J Comp Physiol Psychol |volume=47 |issue=6 |pages=419–27 |year=1954 |pmid=13233369 |doi=10.1037/h0058775}} [http://www.wadsworth.com/psychology_d/templates/student_resources/0155060678_rathus/ps/ps02.html article]</ref> In rats, stimulation of the [[ventral tegmental area]] causes the release of dopamine in the nucleus accumbens much in the same way as addictive drugs and natural reinforcers, such as water or food, initiate the release of dopamine in the nucleus accumbens.<ref>Nakahara, D., Ozaki, N., Miura, Y., Miura, H., et al. Increased dopamine and serotonin metabolism in rat nucleus accumbens produced by intracranial sel-stimulation of medial forebrain bundle as measured by in vivo microdialysis. ''Brain Research'''''', 1989 495: 178-181.</ref> The same results have been seen in human subjects in functional imaging studies. For example, increased dopamine concentration is seen in the extracellular fluid of the nucleus accumbens when subjects believed they were being given money{{Citation needed|date=April 2014}}, and increased activation (i.e., increased fMRI BOLD signal-change) was observed among heterosexual males viewing pictures of attractive women.<ref>Aharon, L., Etcoff, N., Ariely, D., CHabris, C. F., et al. Beautiful faces have variable reward value: fMRI and behavioral evidence. ''Neuron'' 2001, ''32'', 357-551.</ref> |
||
| ⚫ | |||
| ⚫ | An fMRI study conducted in 2005 found that when mother rats were in the presence of their pups the regions of the brain involved in reinforcement, including the nucleus accumbens, were highly active.<ref>Ferris, C.F., Kulkarni, P., Sullivan, J.M., Harder, J.A., et al. Pup sucking is more rewarding than cocaine: Evidence from functional magnetic resonance imaging and three-dimensional computational analysis. ''Journal of Neuroscience'''''', 2005 25: 149-156.</ref> Levels of dopamine increase in the nucleus accumbens during maternal behavior, while lesions in this area upset maternal behavior.<ref>Numan, M. Motivational systems and the neural circuitry of maternal behavior in the rat. ''Developmental Psychobiology'''''', 2007 49: 12-21.</ref> When human mothers are presented pictures of their children, fMRIs show an increased brain activity in the nucleus accumbens and other reinforcing brain regions and a decrease in activity in areas of the brain involved with negative emotions.{{citation needed|date=June 2013}} |
||
=== |
===Depression=== |
||
In April 2007, two research teams reported on having inserted electrodes into the nucleus accumbens in order to use [[deep brain stimulation]] to treat severe [[clinical depression|depression]].<ref>[http://www.technologyreview.com/Biotech/18590/ Brain Electrodes Help Treat Depression], ''[[Technology Review]]'', 26 April 2007</ref> In 2010 experiments reported that deep brain stimulation of the nucleus accumbens was successful in decreasing depression symptoms in 50% of patients who did not respond to other treatments such as [[electroconvulsive therapy]].<ref>Bewernick, B. H., Hurlemann, R., Matusch, A., et al. Nucleus accumbens deep brain stimulation decreases ratings of depression and anciety in treatment-resistant depression. ''Biological Psychiatry'''''', 2009 67: 110-116.</ref> Nucleus accumbens has also been used as a target to treat small groups of patients with therapy-refractory obsessive-compulsive disorder.<ref>Ooms P, Mantione M, Figee M, Schuurman PR, van den Munckhof P, Denys D. Deep brain stimulation for obsessive-compulsive disorders: long-term analysis of quality of life. J Neurol Neurosurg Psychiatry. 2014;85(2):153-8.</ref> |
In April 2007, two research teams reported on having inserted electrodes into the nucleus accumbens in order to use [[deep brain stimulation]] to treat severe [[clinical depression|depression]].<ref>[http://www.technologyreview.com/Biotech/18590/ Brain Electrodes Help Treat Depression], ''[[Technology Review]]'', 26 April 2007</ref> In 2010 experiments reported that deep brain stimulation of the nucleus accumbens was successful in decreasing depression symptoms in 50% of patients who did not respond to other treatments such as [[electroconvulsive therapy]].<ref>Bewernick, B. H., Hurlemann, R., Matusch, A., et al. Nucleus accumbens deep brain stimulation decreases ratings of depression and anciety in treatment-resistant depression. ''Biological Psychiatry'''''', 2009 67: 110-116.</ref> Nucleus accumbens has also been used as a target to treat small groups of patients with therapy-refractory obsessive-compulsive disorder.<ref>Ooms P, Mantione M, Figee M, Schuurman PR, van den Munckhof P, Denys D. Deep brain stimulation for obsessive-compulsive disorders: long-term analysis of quality of life. J Neurol Neurosurg Psychiatry. 2014;85(2):153-8.</ref> |
||
Revision as of 08:08, 11 October 2014
| Nucleus accumbens | |
|---|---|
| File:Circuit du système de recompense.jpg Nucleus accumbens visible in red | |
 Medial surface, person facing to the left. Nucleus accumbens is very roughly in Brodmann area 34 | |
| Details | |
| Part of | Mesolimbic pathway Basal ganglia (Ventral striatum) |
| Parts | Nucleus accumbens shell Nucleus accumbens core |
| Identifiers | |
| Latin | nucleus accumbens septi |
| Acronym(s) | NAc or NAcc |
| MeSH | D009714 |
| NeuroNames | 277 |
| NeuroLex ID | birnlex_727 |
| TA98 | A14.1.09.440 |
| TA2 | 5558 |
| FMA | 61889 |
| Anatomical terms of neuroanatomy | |
The nucleus accumbens (NAc or NAcc), also known as the accumbens nucleus or as the nucleus accumbens septi (Latin for nucleus adjacent to the septum) is a region in the basal forebrain rostral to the preoptic area of the hypothalamus.[1] The nucleus accumbens and the olfactory tubercle collectively form the ventral striatum, which is part of the basal ganglia.[2] Nucleus accumbens has a significant role in cognitive processing involving motivation, pleasure, and reward and reinforcement learning, and hence has significant role in addiction.[3][4] It plays a lesser role in fear, impulsivity, and the placebo effect.[5][6][7] It is involved in the encoding of new motor programs as well.[3]
Each cerebral hemisphere has its own nucleus accumbens. It is located where the head of the caudate and the anterior portion of the putamen meet just lateral to the septum pellucidum. The nucleus accumbens can be divided into two structures—the nucleus accumbens core and the nucleus accumbens shell. These structures have different morphology and function.
Structure
The nucleus accumbens is an aggregate of neurons which is described as having an outer shell and an inner core.[3]
Cell types
The core of the NAcc is made up mainly of medium spiny neurons. Compared to the neurons in the shell, those in the core have an increased density of dendritic spines, branch segments, and terminal segments. From the core, the neurons project to other sub-cortical areas such as the globus pallidus and the substantia nigra. GABA is one of the main neurotransmitters in the NAcc, and GABA receptors are also abundant.[8][9] These neurons are also the main projection or output neurons of the nucleus accumbens.
While 95% of the neurons projecting from the nucleus accumbens are medium spiny GABA-ergic neurons, other projecting neuronal types are also present, such as large cholinergic interneurons.
Output
The output neurons of the nucleus accumbens send axon projections to the basal ganglia and the ventral analog of the globus pallidus, known as the ventral pallidum (VP). The VP, in turn, projects to the medial dorsal nucleus of the dorsal thalamus, which projects to the prefrontal cortex as well as the striatum. Other efferents from the nucleus accumbens include connections with the substantia nigra, and the reticular formation of the pons.[1]
Input
Major inputs to the nucleus accumbens include prefrontal association cortices, basolateral amygdala, and dopaminergic neurons located in the ventral tegmental area (VTA), which connect via the mesolimbic pathway. Thus the nucleus accumbens is often described as one part of a cortico-striato-thalamo-cortical loop.
Dopaminergic input from the VTA modulate the activity of neurons within the nucleus accumbens. These neurons are activated directly or indirectly by euphoriant drugs (e.g., amphetamine, opiates, nicotine, etc.) and by participating in rewarding experiences (e.g., sex, music, exercise, etc.).[10][11]
Another major source of input comes from the CA1 and ventral subiculum of the hippocampus to the dorsomedial area of the nucleus accumbens. The neurons of the hippocampus have a noteworthy correlation to slight depolarizations of cells in the nucleus accumbens, which makes them more positive and therefore more excitable. The correlated cells of these excited states of the medium spiny neurons in the nucleus accumbens are shared equally between the subiculum and CA1. The subiculum neurons are found to hyperpolarize (increase negativity) while the CA1 neurons "ripple" (fire > 50 Hz) in order to accomplish this priming.[12]
Shell
The nucleus accumbens shell is a substructure of the nucleus accumbens. The shell and core together form the entire nucleus accumbens.
Location: The shell is the outer region of the nucleus accumbens, and – unlike the core – is considered to be part of the extended amygdala, located at its rostral pole.
Cell types: Neurons in the nucleus accumbens are mostly medium spiny neurons. The neurons in the shell, as compared to the core, have a lower density of dendritic spines, less terminal segments, and less branch segments than those in the core. The shell neurons project to the subcommissural part of the ventral pallidum as well as the ventral tegmental area and to extensive areas in the hypothalamus and extended amygdala.[13][14][15]
Function: The shell of the nucleus accumbens is involved in the cognitive processing of motivational salience (wanting) as well as reward and reinforcement effects.[3] Particularly important are the effects of drug and naturally rewarding stimuli on the NAc shell because these effects are related to addiction.[3] Addictive drugs have a larger effect on dopamine release in the shell than in the core.[3]
Core
The nucleus accumbens core is the inner substructure of the nucleus accumbens.
Location: The nucleus accumbens core is part of the ventral striatum, located within the basal ganglia.
Cell types: The core of the NAcc is made up mainly of medium spiny neurons. The neurons in the core, as compared to the neurons in the shell, have an increased density of dendritic spines, branch segments, and terminal segments. From the core, the neurons project to other sub-cortical areas such as the globus pallidus and the substantia nigra. GABA is one of the main neurotransmitters in the NAcc, and GABA receptors are also abundant.[8][16]
Function: The nucleus accumbens core is involved in the cognitive processing of motor function related to reward and reinforcement.[3] Specifically, the core encodes new motor programs which facilitate the acquisition of a given reward in the future.[3]
Cell types
The core of the NAcc is made up mainly of medium spiny neurons. The neurons in the core, as compared to the neurons in the shell, have an increased density of dendritic spines, branch segments, and terminal segments. From the core, the neurons project to other sub-cortical areas such as the globus pallidus and the substantia nigra. GABA is one of the main neurotransmitters in the NAcc, and GABA receptors are also abundant.[8][17]
Neurotransmitters
Dopamine: Dopamine is related to recreational drugs including amphetamines, cocaine, and morphine, which increase extracellular levels of dopamine in both the NAc shell and the NAc core, but the effect of these increases is more pronounced in the shell. Only amphetamine at high levels increased extracellular levels of dopamine to similar levels in both the shell and the core. All of this points to a 'functional heterogeneity' in the nucleus accumbens between the shell region and the core region.[18] Similarly to drug rewards, non-drug rewards also increase levels of extracellular dopamine in the NAc shell, but drug induced DA increase is more resilient to habituation when exposed repeatedly to drug-stimuli, unlike non-drug rewarding stimuli induced dopamine increases, which do succumb to habituation. Recent[when?] studies have shown that the repeated influence of drug-inducing DA projection has an abnormal strengthening effect on stimulus-drug associations and increases the drug-reward stimuli’s resistance to extinction. This may be a contributing factor to addiction. This effect was more pronounced in the NAc shell than in the NAc core.[13][13][19]
Glucocorticoids and dopamine:Glucocorticoid receptors are the only corticosteroid receptors in the nucleus accumbens shell. L-DOPA, steroids, and specifically glucocorticoids are currently known to be the only known endogenous compounds that can induce psychotic problems, so understanding the hormonal control over dopaminergic projections with regards to glucocorticoid receptors could lead to new treatments for psychotic symptoms. A recent study demonstrated that suppression of the glucocorticoid receptors led to a decrease in the release of dopamine, which may lead to future research involving anti-glucocorticoid drugs to potentially relieve psychotic symptoms.[20]
GABA: A recent study on rats that used GABA agonists and antagonists indicated that GABAA receptors in the NAc shell have inhibitory control on turning behavior influenced by dopamine, and GABAB receptors have inhibitory control over turning behavior mediated by acetylcholine.[13][21]
Glutamate: Studies have shown that local blockade of glutamatergic NMDA receptors in the NAcc core impaired spatial learning, but did not have an effect in the NAcc shell.[22] Another study demonstrated that both NMDA and AMPA (both glutamate receptors) play important roles in regulating instrumental learning.[23]
Serotonin (5-HT): Overall, 5-HT synapses are more abundant and have a greater number of synaptic contacts in the NAc shell than in the core. They are also larger and thicker, and contain more large dense core vesicles than their counterparts in the core.
Function
Pleasure and reinforcement
Although the nucleus accumbens has traditionally been studied for its role in addiction, it plays an equal role in processing many rewards such as food and sex. The nucleus accumbens is selectively activated during the perception of pleasant, emotionally arousing pictures and during mental imagery of pleasant, emotional scenes.[24][25] A 2005 study found that it is involved in the regulation of emotions induced by music,[26] perhaps consequent to its role in mediating dopamine release. The nucleus accumbens plays a role in rhythmic timing and is considered to be of central importance to the limbic-motor interface (Mogensen).[citation needed]
In the 1950s, James Olds and Peter Milner implanted electrodes into the septal area of the rat and found that the rat chose to press a lever which stimulated it. It continued to prefer this even over stopping to eat or drink. This suggests that the area is the "pleasure center" of the brain and is involved in reinforcement learning.[27] In rats, stimulation of the ventral tegmental area causes the release of dopamine in the nucleus accumbens much in the same way as addictive drugs and natural reinforcers, such as water or food, initiate the release of dopamine in the nucleus accumbens.[28] The same results have been seen in human subjects in functional imaging studies. For example, increased dopamine concentration is seen in the extracellular fluid of the nucleus accumbens when subjects believed they were being given money[citation needed], and increased activation (i.e., increased fMRI BOLD signal-change) was observed among heterosexual males viewing pictures of attractive women.[29]
Maternal behavior
An fMRI study conducted in 2005 found that when mother rats were in the presence of their pups the regions of the brain involved in reinforcement, including the nucleus accumbens, were highly active.[30] Levels of dopamine increase in the nucleus accumbens during maternal behavior, while lesions in this area upset maternal behavior.[31] When human mothers are presented pictures of their children, fMRIs show an increased brain activity in the nucleus accumbens and other reinforcing brain regions and a decrease in activity in areas of the brain involved with negative emotions.[citation needed]
Clinical significance
Addiction
Current models of addiction from chronic drug use involve alterations in gene expression in the mesocorticolimbic projection.[10][32][33] The most important transcription factors that produce these alterations are ΔFosB, cyclic adenosine monophosphate (cAMP) response element binding protein (CREB), and nuclear factor kappa B (NFκB).[10] ΔFosB is the most significant gene transcription factor in addiction since its viral or genetic overexpression in the nucleus accumbens is necessary and sufficient for many of the neural adaptations seen in drug addiction;[10] it has been implicated in addictions to alcohol, cannabinoids, cocaine, nicotine, phenylcyclidine, opiates, and substituted amphetamines.[10][32][34] ΔJunD is the transcription factor which directly opposes ΔFosB.[10] Increases in nucleus accumbens ΔJunD expression can reduce or, with a large increase, even block most of the neural alterations seen in chronic drug abuse (i.e., the alterations mediated by ΔFosB).[10]
ΔFosB also plays an important role in regulating behavioral responses to natural rewards, such as palatable food, sex, and exercise.[10][11] Natural rewards, like drugs of abuse, induce ΔFosB in the nucleus accumbens, and chronic acquisition of these rewards can result in a similar pathological addictive state through ΔFosB overexpression.[10][11][35] Consequently, ΔFosB is the key transcription factor involved in addictions to natural rewards as well;[10][11][35] in particular, ΔFosB in the nucleus accumbens is critical for the reinforcing effects of sexual reward.[11] Research on the interaction between natural and drug rewards suggests that psychostimulants and sexual behavior act on similar biomolecular mechanisms to induce ΔFosB in the nucleus accumbens and possess cross-sensitization effects that are mediated through ΔFosB.[36][35]
| nucleus accumbens | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | Nucleus accumbensNAcc | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | GeneCards: [1]; OMA:- orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Protein fosB, also known as FosB and G0/G1 switch regulatory protein 3 (G0S3), is a protein that in humans is encoded by the FBJ murine osteosarcoma viral oncogene homolog B (FOSB) gene.[37][38][39]
The FOS gene family consists of four members: FOS, FOSB, FOSL1, and FOSL2. These genes encode leucine zipper proteins that can dimerize with proteins of the JUN family (e.g., c-Jun, JunD), thereby forming the transcription factor complex AP-1. As such, the FOS proteins have been implicated as regulators of cell proliferation, differentiation, and transformation.[37] FosB and its truncated splice variants, ΔFosB and further truncated Δ2ΔFosB, are all involved in osteosclerosis, although Δ2ΔFosB lacks a known transactivation domain, in turn preventing it from affecting transcription through the AP-1 complex.[40]
The ΔFosB splice variant has been identified as playing a central, crucial[41][10] role in the development and maintenance of addiction.[41][35][42] ΔFosB overexpression (i.e., an abnormally and excessively high level of ΔFosB expression which produces a pronounced gene-related phenotype) triggers the development of addiction-related neuroplasticity throughout the reward system and produces a behavioral phenotype that is characteristic of an addiction.[41][42][43] ΔFosB differs from the full length FosB and further truncated Δ2ΔFosB in its capacity to produce these effects, as only accumbal ΔFosB overexpression is associated with pathological responses to drugs.[44]
DeltaFosB
DeltaFosB – more commonly written as ΔFosB – is a truncated splice variant of the FOSB gene.[45] ΔFosB has been implicated as a critical factor in the development of virtually all forms of behavioral and drug addictions.[10][35][11] In the brain's reward system, it is linked to changes in a number of other gene products, such as CREB and sirtuins.[46][47][48] In the body, ΔFosB regulates the commitment of mesenchymal precursor cells to the adipocyte or osteoblast lineage.[49]
In the nucleus accumbens, ΔFosB functions as a "sustained molecular switch" and "master control protein" in the development of an addiction.[41][50][51] In other words, once "turned on" (sufficiently overexpressed) ΔFosB triggers a series of transcription events that ultimately produce an addictive state (i.e., compulsive reward-seeking involving a particular stimulus); this state is sustained for months after cessation of drug use due to the abnormal and exceptionally long half-life of ΔFosB isoforms.[41][50][51] ΔFosB expression in D1-type nucleus accumbens medium spiny neurons directly and positively regulates drug self-administration and reward sensitization through positive reinforcement while decreasing sensitivity to aversion.[41][42] Based upon the accumulated evidence, a medical review from late 2014 argued that accumbal ΔFosB expression can be used as an addiction biomarker and that the degree of accumbal ΔFosB induction by a drug is a metric for how addictive it is relative to others.[41]
Chronic administration of anandamide, or N-arachidonylethanolamide (AEA), an endogenous cannabinoid, and additives such as sucralose, a noncaloric sweetener used in many food products of daily intake, are found to induce an overexpression of ΔFosB in the infralimbic cortex (Cx), nucleus accumbens (NAc) core, shell, and central nucleus of amygdala (Amy), that induce long-term changes in the reward system.[52]
Role in addiction
| Addiction and dependence glossary[42][53][54] | |
|---|---|
| |
Chronic addictive drug use causes alterations in gene expression in the mesocorticolimbic projection, which arise through transcriptional and epigenetic mechanisms.[10][32][33] The most important transcription factors that produce these alterations are ΔFosB, cyclic adenosine monophosphate (cAMP) response element binding protein (CREB), and nuclear factor kappa B (NF-κB).[10] ΔFosB is the most significant biomolecular mechanism in addiction because the overexpression of ΔFosB in the D1-type medium spiny neurons in the nucleus accumbens is necessary and sufficient for many of the neural adaptations and behavioral effects (e.g., expression-dependent increases in drug self-administration and reward sensitization) seen in drug addiction.[41][10][42] ΔFosB overexpression has been implicated in addictions to alcohol, cannabinoids, cocaine, methylphenidate, nicotine, opioids, phencyclidine, propofol, and substituted amphetamines, among others.[41][10][32][34][61] ΔJunD, a transcription factor, and G9a, a histone methyltransferase, both oppose the function of ΔFosB and inhibit increases in its expression.[10][42][62] Increases in nucleus accumbens ΔJunD expression (via viral vector-mediated gene transfer) or G9a expression (via pharmacological means) reduces, or with a large increase can even block, many of the neural and behavioral alterations seen in chronic drug abuse (i.e., the alterations mediated by ΔFosB).[43][10] Repression of c-Fos by ΔFosB, which consequently further induces expression of ΔFosB, forms a positive feedback loop that serves to indefinitely perpetuate the addictive state.
ΔFosB also plays an important role in regulating behavioral responses to natural rewards, such as palatable food, sex, and exercise.[10][11] Natural rewards, similar to drugs of abuse, induce gene expression of ΔFosB in the nucleus accumbens, and chronic acquisition of these rewards can result in a similar pathological addictive state through ΔFosB overexpression.[10][35][11] Consequently, ΔFosB is the key mechanism involved in addictions to natural rewards (i.e., behavioral addictions) as well;[10][35][11] in particular, ΔFosB in the nucleus accumbens is critical for the reinforcing effects of sexual reward.[11] Research on the interaction between natural and drug rewards suggests that dopaminergic psychostimulants (e.g., amphetamine) and sexual behavior act on similar biomolecular mechanisms to induce ΔFosB in the nucleus accumbens and possess bidirectional reward cross-sensitization effects[note 1] that are mediated through ΔFosB.[35][36] This phenomenon is notable since, in humans, a dopamine dysregulation syndrome, characterized by drug-induced compulsive engagement in natural rewards (specifically, sexual activity, shopping, and gambling), has also been observed in some individuals taking dopaminergic medications.[35]
ΔFosB inhibitors (drugs or treatments that oppose its action or reduce its expression) may be an effective treatment for addiction and addictive disorders.[63] Current medical reviews of research involving lab animals have identified a drug class – class I histone deacetylase inhibitors[note 2] – that indirectly inhibits the function and further increases in the expression of accumbal ΔFosB by inducing G9a expression in the nucleus accumbens after prolonged use.[43][62][64][65] These reviews and subsequent preliminary evidence which used oral administration or intraperitoneal administration of the sodium salt of butyric acid or other class I HDAC inhibitors for an extended period indicate that these drugs have efficacy in reducing addictive behavior in lab animals[note 3] that have developed addictions to ethanol, psychostimulants (i.e., amphetamine and cocaine), nicotine, and opiates;[62][65][66][67] however, as of August 2015[update], few clinical trials involving humans with addiction and any HDAC class I inhibitors have been conducted to test for treatment efficacy in humans or identify an optimal dosing regimen.[note 4]
Plasticity in cocaine addiction
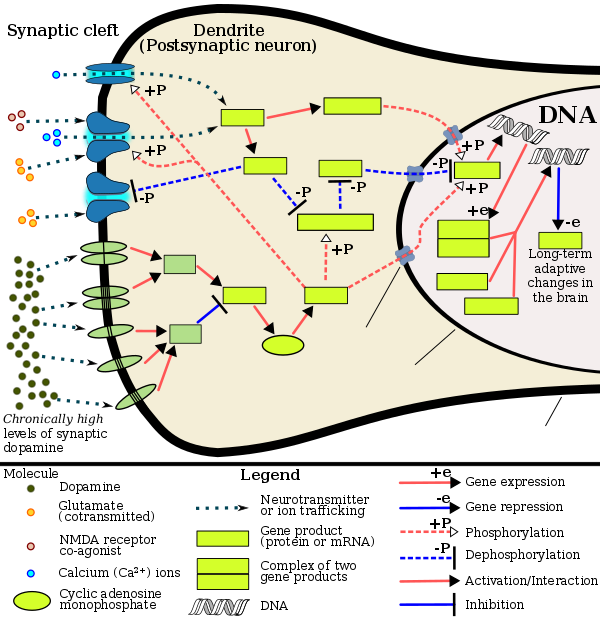
ΔFosB accumulation from excessive drug use
Top: this depicts the initial effects of high dose exposure to an addictive drug on gene expression in the nucleus accumbens for various Fos family proteins (i.e., c-Fos, FosB, ΔFosB, Fra1, and Fra2).
Bottom: this illustrates the progressive increase in ΔFosB expression in the nucleus accumbens following repeated twice daily drug binges, where these phosphorylated (35–37 kilodalton) ΔFosB isoforms persist in the D1-type medium spiny neurons of the nucleus accumbens for up to 2 months.[51][59] |
ΔFosB levels have been found to increase upon the use of cocaine.[69] Each subsequent dose of cocaine continues to increase ΔFosB levels with no apparent ceiling of tolerance.[citation needed] Elevated levels of ΔFosB leads to increases in brain-derived neurotrophic factor (BDNF) levels, which in turn increases the number of dendritic branches and spines present on neurons involved with the nucleus accumbens and prefrontal cortex areas of the brain. This change can be identified rather quickly, and may be sustained weeks after the last dose of the drug.
Transgenic mice exhibiting inducible expression of ΔFosB primarily in the nucleus accumbens and dorsal striatum exhibit sensitized behavioural responses to cocaine.[70] They self-administer cocaine at lower doses than control,[71] but have a greater likelihood of relapse when the drug is withheld.[51][71] ΔFosB increases the expression of AMPA receptor subunit GluR2[70] and also decreases expression of dynorphin, thereby enhancing sensitivity to reward.[51]
| Target gene |
Target expression |
Neural effects | Behavioral effects |
|---|---|---|---|
| c-Fos | ↓ | Molecular switch enabling the chronic induction of ΔFosB[note 5] |
– |
| dynorphin | ↓ [note 6] |
• Downregulation of κ-opioid feedback loop | • Diminished self-extinguishing response to drug |
| NF-κB | ↑ | • Expansion of Nacc dendritic processes • NF-κB inflammatory response in the NAcc • NF-κB inflammatory response in the CP |
• Increased drug reward • Locomotor sensitization |
| GluR2 | ↑ | • Decreased sensitivity to glutamate | • Increased drug reward |
| Cdk5 | ↑ | • GluR1 synaptic protein phosphorylation • Expansion of NAcc dendritic processes |
• Decreased drug reward (net effect) |
Summary of addiction-related plasticity
| Form of neuroplasticity or behavioral plasticity |
Type of reinforcer | Sources | |||||
|---|---|---|---|---|---|---|---|
| Opiates | Psychostimulants | High fat or sugar food | Sexual intercourse | Physical exercise (aerobic) |
Environmental enrichment | ||
| ΔFosB expression in nucleus accumbens D1-type MSNs |
↑ | ↑ | ↑ | ↑ | ↑ | ↑ | [35] |
| Behavioral plasticity | |||||||
| Escalation of intake | Yes | Yes | Yes | [35] | |||
| Psychostimulant cross-sensitization |
Yes | Not applicable | Yes | Yes | Attenuated | Attenuated | [35] |
| Psychostimulant self-administration |
↑ | ↑ | ↓ | ↓ | ↓ | [35] | |
| Psychostimulant conditioned place preference |
↑ | ↑ | ↓ | ↑ | ↓ | ↑ | [35] |
| Reinstatement of drug-seeking behavior | ↑ | ↑ | ↓ | ↓ | [35] | ||
| Neurochemical plasticity | |||||||
| CREB phosphorylation in the nucleus accumbens |
↓ | ↓ | ↓ | ↓ | ↓ | [35] | |
| Sensitized dopamine response in the nucleus accumbens |
No | Yes | No | Yes | [35] | ||
| Altered striatal dopamine signaling | ↓DRD2, ↑DRD3 | ↑DRD1, ↓DRD2, ↑DRD3 | ↑DRD1, ↓DRD2, ↑DRD3 | ↑DRD2 | ↑DRD2 | [35] | |
| Altered striatal opioid signaling | No change or ↑μ-opioid receptors |
↑μ-opioid receptors ↑κ-opioid receptors |
↑μ-opioid receptors | ↑μ-opioid receptors | No change | No change | [35] |
| Changes in striatal opioid peptides | ↑dynorphin No change: enkephalin |
↑dynorphin | ↓enkephalin | ↑dynorphin | ↑dynorphin | [35] | |
| Mesocorticolimbic synaptic plasticity | |||||||
| Number of dendrites in the nucleus accumbens | ↓ | ↑ | ↑ | [35] | |||
| Dendritic spine density in the nucleus accumbens |
↓ | ↑ | ↑ | [35] | |||
Other functions in the brain
Viral overexpression of ΔFosB in the output neurons of the nigrostriatal dopamine pathway (i.e., the medium spiny neurons in the dorsal striatum) induces levodopa-induced dyskinesias in animal models of Parkinson's disease.[72][73] Dorsal striatal ΔFosB is overexpressed in rodents and primates with dyskinesias;[73] postmortem studies of individuals with Parkinson's disease that were treated with levodopa have also observed similar dorsal striatal ΔFosB overexpression.[73] Levetiracetam, an antiepileptic drug, has been shown to dose-dependently decrease the induction of dorsal striatal ΔFosB expression in rats when co-administered with levodopa;[73] the signal transduction involved in this effect is unknown.[73]
ΔFosB expression in the nucleus accumbens shell increases resilience to stress and is induced in this region by acute exposure to social defeat stress.[74][75][76]
Antipsychotic drugs have been shown to increase ΔFosB as well, more specifically in the prefrontal cortex. This increase has been found to be part of pathways for the negative side effects that such drugs produce.[77]
See also
Notes
- ^ In simplest terms, this means that when either amphetamine or sex is perceived as "more alluring or desirable" through reward sensitization, this effect occurs with the other as well.
- ^ Inhibitors of class I histone deacetylase (HDAC) enzymes are drugs that inhibit four specific histone-modifying enzymes: HDAC1, HDAC2, HDAC3, and HDAC8. Most of the animal research with HDAC inhibitors has been conducted with four drugs: butyrate salts (mainly sodium butyrate), trichostatin A, valproic acid, and SAHA;[64][65] butyric acid is a naturally occurring short-chain fatty acid in humans, while the latter two compounds are FDA-approved drugs with medical indications unrelated to addiction.
- ^ Specifically, prolonged administration of a class I HDAC inhibitor appears to reduce an animal's motivation to acquire and use an addictive drug without affecting an animals motivation to attain other rewards (i.e., it does not appear to cause motivational anhedonia) and reduce the amount of the drug that is self-administered when it is readily available.[62][65][66]
- ^ Among the few clinical trials that employed a class I HDAC inhibitor, one utilized valproate for methamphetamine addiction.[68]
- ^ In other words, c-Fos repression allows ΔFosB to accumulate within nucleus accumbens medium spiny neurons more rapidly because it is selectively induced in this state.[42]
- ^ ΔFosB has been implicated in causing both increases and decreases in dynorphin expression in different studies;[41][46] this table entry reflects only a decrease.
- Image legend
- ^ (Text color) Transcription factors
References
- ^ a b Carlson, Neil R. Physiology of Behavior. 11th ed. Boston: Pearson, 2013. Print.
- ^ Nucleus Accumbens
- ^ a b c d e f g h Malenka RC, Nestler EJ, Hyman SE (2009). "Chapter 15: Reinforcement and Addictive Disorders". In Sydor A, Brown RY (ed.). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. pp. 147–148, 367, 376. ISBN 9780071481274.
VTA DA neurons play a critical role in motivation, reward-related behavior (Chapter 15), attention, and multiple forms of memory. This organization of the DA system, wide projection from a limited number of cell bodies, permits coordinated responses to potent new rewards. Thus, acting in diverse terminal fields, dopamine confers motivational salience ("wanting") on the reward itself or associated cues (nucleus accumbens shell region), updates the value placed on different goals in light of this new experience (orbital prefrontal cortex), helps consolidate multiple forms of memory (amygdala and hippocampus), and encodes new motor programs that will facilitate obtaining this reward in the future (nucleus accumbens core region and dorsal striatum). In this example, dopamine modulates the processing of sensorimotor information in diverse neural circuits to maximize the ability of the organism to obtain future rewards. ...
The brain reward circuitry that is targeted by addictive drugs normally mediates the pleasure and strengthening of behaviors associated with natural reinforcers, such as food, water, and sexual contact. Dopamine neurons in the VTA are activated by food and water, and dopamine release in the NAc is stimulated by the presence of natural reinforcers, such as food, water, or a sexual partner. ...
The NAc and VTA are central components of the circuitry underlying reward and memory of reward. As previously mentioned, the activity of dopaminergic neurons in the VTA appears to be linked to reward prediction. The NAc is involved in learning associated with reinforcement and the modulation of motoric responses to stimuli that satisfy internal homeostatic needs. The shell of the NAc appears to be particularly important to initial drug actions within reward circuitry; addictive drugs appear to have a greater effect on dopamine release in the shell than in the core of the NAc.{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Malenka RC, Nestler EJ, Hyman SE (2009). "Chapter 10: Neural and Neuroendocrine Control of the Internal Milieu". In Sydor A, Brown RY (ed.). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. p. 266. ISBN 9780071481274.
Dopamine acts in the nucleus accumbens to attach motivational significance to stimuli associated with reward.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Schwienbacher I, Fendt M, Richardson R, Schnitzler HU (2004). "Temporary inactivation of the nucleus accumbens disrupts acquisition and expression of fear-potentiated startle in rats". Brain Res. 1027 (1–2): 87–93. doi:10.1016/j.brainres.2004.08.037. PMID 15494160.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Zubieta JK, Stohler CS (2009). "Neurobiological mechanisms of placebo responses". Ann. N. Y. Acad. Sci. 1156: 198–210. doi:10.1111/j.1749-6632.2009.04424.x. PMC 3073412. PMID 19338509.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Basar K, Sesia T, Groenewegen H, Steinbusch HW, Visser-Vandewalle V, Temel Y (2010). "Nucleus accumbens and impulsivity". Prog. Neurobiol. 92 (4): 533–57. doi:10.1016/j.pneurobio.2010.08.007. PMID 20831892.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b c Shirayama, Yukihiko, and Shigeyuki Chaki. "Neurochemistry of the Nucleus Accumbens and Its Relevance to Depression and Antidepressant Action in Rodents." Current Neuropharmocology 4.4 (2006): 277-91. Bentham Science Publishers Ltd. Web. 16 Nov. 2011. <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2475798/>.
- ^ Meredith, G. E., C. M. Pennartz, and H. J. Groenewegen. "The Cellular Framework for Chemical Signalling in the Nucleus Accumbens." Progress in Brain Research 99 (1993): 3-24. Web. 16 Nov. 2011. <http://www.ncbi.nlm.nih.gov/pubmed/7906426>.
- ^ a b c d e f g h i j k l m n o p q r s t u Robison AJ, Nestler EJ (November 2011). "Transcriptional and epigenetic mechanisms of addiction". Nat. Rev. Neurosci. 12 (11): 623–637. doi:10.1038/nrn3111. PMC 3272277. PMID 21989194.
ΔFosB has been linked directly to several addiction-related behaviors ... Importantly, genetic or viral overexpression of ΔJunD, a dominant negative mutant of JunD which antagonizes ΔFosB- and other AP-1-mediated transcriptional activity, in the NAc or OFC blocks these key effects of drug exposure14,22–24. This indicates that ΔFosB is both necessary and sufficient for many of the changes wrought in the brain by chronic drug exposure. ΔFosB is also induced in D1-type NAc MSNs by chronic consumption of several natural rewards, including sucrose, high fat food, sex, wheel running, where it promotes that consumption14,26–30. This implicates ΔFosB in the regulation of natural rewards under normal conditions and perhaps during pathological addictive-like states.
Cite error: The named reference "Nestler" was defined multiple times with different content (see the help page). - ^ a b c d e f g h i j Blum K, Werner T, Carnes S, Carnes P, Bowirrat A, Giordano J, Oscar-Berman M, Gold M (2012). "Sex, drugs, and rock 'n' roll: hypothesizing common mesolimbic activation as a function of reward gene polymorphisms". J. Psychoactive Drugs. 44 (1): 38–55. PMC 4040958. PMID 22641964.
{{cite journal}}: CS1 maint: multiple names: authors list (link) Cite error: The named reference "ΔFosB reward" was defined multiple times with different content (see the help page). - ^ O'Donnell, P., Goto, Y. (2001). "Synchronous activity in the hippocampus and nucleus accumbens in vivo". J. Neurosci. 21 (4): RC131. PMID 11160416.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c d Shirayama, Y; Chaki, S (2006). "Neurochemistry of the Nucleus Accumbens and its Relevance to Depression and Antidepressant Action in Rodents". Current Neuropharmacology. 4 (4): 277–291. doi:10.2174/157015906778520773. PMC 2475798. PMID 18654637.
- ^ Meredith, GE; Agolia, R; Arts, MP; Groenewegen, HJ; Zahm, DS (1992). "Morphological differences between projection neurons of the core and shell in the nucleus accumbens of the rat". Neuroscience. 50 (1): 149–62. doi:10.1016/0306-4522(92)90389-j. PMID 1383869.
- ^ Meredith, GE; Pennartz, CM; Groenewegen, HJ (1993). "The cellular framework for chemical signalling in the nucleus accumbens". Progress in brain research. 99: 3–24. doi:10.1016/s0079-6123(08)61335-7. PMID 7906426.
- ^ Meredith, G. E., C. M. Pennartz, and H. J. Groenewegen. "The Cellular Framework for Chemical Signalling in the Nucleus Accumbens." Progress in Brain Research 99 (1993): 3-24. Web. 16 Nov. 2011. <http://www.ncbi.nlm.nih.gov/pubmed/7906426>.
- ^ Meredith, G. E., C. M. Pennartz, and H. J. Groenewegen. "The Cellular Framework for Chemical Signalling in the Nucleus Accumbens." Progress in Brain Research 99 (1993): 3-24. Web. 16 Nov. 2011. <http://www.ncbi.nlm.nih.gov/pubmed/7906426>.
- ^ Pontieri, FE; Tanda, G; Di Chiara, G (1995). "Intravenous cocaine, morphine, and amphetamine preferentially increase extracellular dopamine in the "shell" as compared with the "core" of the rat nucleus accumbens". Proceedings of the National Academy of Sciences of the United States of America. 92 (26): 12304–12308. doi:10.1073/pnas.92.26.12304. PMC 40345. PMID 8618890.
- ^ Di Chiara, G (2002). "Nucleus accumbens shell and core dopamine: Differential role in behavior and addiction". Behavioural Brain Research. 137 (1–2): 75–114. doi:10.1016/s0166-4328(02)00286-3. PMID 12445717.
- ^ Barrot, M; Marinelli, M; Abrous, DN; Rougé-Pont, F; Le Moal, M; Piazza, PV (2000). "The dopaminergic hyper-responsiveness of the shell of the nucleus accumbens is hormone-dependent" (PDF). The European Journal of Neuroscience. 12 (3): 973–9. doi:10.1046/j.1460-9568.2000.00996.x. PMID 10762327.
- ^ Akiyama, G; Ikeda, H; Matsuzaki, S; Sato, M; Moribe, S; Koshikawa, N; Cools, AR (2004). "GABAA and GABAB receptors in the nucleus accumbens shell differentially modulate dopamine and acetylcholine receptor-mediated turning behaviour". Neuropharmacology. 46 (8): 1082–8. doi:10.1016/j.neuropharm.2004.02.007. PMID 15111014.
- ^ Smith-Roe, S. L., K. Sadeghian, and A. E. Kelley. "Spatial Learning and Performance in the Radial Arm Maze Is Impaired after N-methyl-D-aspartate (NMDA) Receptor Blockade in Striatal Subregions." Behav Neurosci. 113.4 (1999): 703-17. Web. 16 Nov. 2011. <http://www.ncbi.nlm.nih.gov/pubmed/10495079>.
- ^ Giertler, C., I. Bohn, and W. Hauber. "Involvement of NMDA and AMPA/KA Receptors in the Nucleus Accumbens Core in Instrumental Learning Guided by Reward-predictive Cues." Eur J Neurosci. 21.6 (2005): 1689-702. Web. 16 Nov. 2011. <http://www.ncbi.nlm.nih.gov/pubmed/15845096>.
- ^ Costa, VD, Lang, PJ, Sabatinelli, D, Bradley MM, and Versace, F (2010). "Emotional imagery: Assessing pleasure and arousal in the brain's reward circuitry". Human Brain Mapping. 31 (9): 1446–1457. doi:10.1002/hbm.20948. PMID 20127869.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Sabatinelli, D, Lang, PJ, Bradley, MM, Costa, VD, and Versace, F (2007). "Pleasure rather than salience activates human nucleus accumbens and medial prefrontal cortex". Journal of Neurophysiology. 98 (9): 1374–1379. doi:10.1152/jn.00230.2007. PMID 17596422.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Menon, Vinod & Levitin, Daniel J. (2005) The rewards of music listening: Response and physiological connectivity of themesolimbic system." NeuroImage 28(1), pp. 175-184
- ^ Olds J, Milner P (1954). "Positive reinforcement produced by electrical stimulation of septal area and other regions of rat brain". J Comp Physiol Psychol. 47 (6): 419–27. doi:10.1037/h0058775. PMID 13233369. article
- ^ Nakahara, D., Ozaki, N., Miura, Y., Miura, H., et al. Increased dopamine and serotonin metabolism in rat nucleus accumbens produced by intracranial sel-stimulation of medial forebrain bundle as measured by in vivo microdialysis. Brain Research', 1989 495: 178-181.
- ^ Aharon, L., Etcoff, N., Ariely, D., CHabris, C. F., et al. Beautiful faces have variable reward value: fMRI and behavioral evidence. Neuron 2001, 32, 357-551.
- ^ Ferris, C.F., Kulkarni, P., Sullivan, J.M., Harder, J.A., et al. Pup sucking is more rewarding than cocaine: Evidence from functional magnetic resonance imaging and three-dimensional computational analysis. Journal of Neuroscience', 2005 25: 149-156.
- ^ Numan, M. Motivational systems and the neural circuitry of maternal behavior in the rat. Developmental Psychobiology', 2007 49: 12-21.
- ^ a b c d Hyman SE, Malenka RC, Nestler EJ (2006). "Neural mechanisms of addiction: the role of reward-related learning and memory". Annu. Rev. Neurosci. 29: 565–598. doi:10.1146/annurev.neuro.29.051605.113009. PMID 16776597.
{{cite journal}}: CS1 maint: multiple names: authors list (link) Cite error: The named reference "Nestler, Hyman, and Malenka 2" was defined multiple times with different content (see the help page). - ^ a b Steiner H, Van Waes V (January 2013). "Addiction-related gene regulation: risks of exposure to cognitive enhancers vs. other psychostimulants". Prog. Neurobiol. 100: 60–80. doi:10.1016/j.pneurobio.2012.10.001. PMC 3525776. PMID 23085425. Cite error: The named reference "Addiction genetics" was defined multiple times with different content (see the help page).
- ^ a b Kanehisa Laboratories (2 August 2013). "Alcoholism – Homo sapiens (human)". KEGG Pathway. Retrieved 10 April 2014. Cite error: The named reference "Alcoholism ΔFosB" was defined multiple times with different content (see the help page).
- ^ a b c d e f g h i j k l m n o p q r s t u v Olsen CM (December 2011). "Natural rewards, neuroplasticity, and non-drug addictions". Neuropharmacology. 61 (7): 1109–1122. doi:10.1016/j.neuropharm.2011.03.010. PMC 3139704. PMID 21459101. Retrieved September 10, 2014.
Cross-sensitization is also bidirectional, as a history of amphetamine administration facilitates sexual behavior and enhances the associated increase in NAc DA ... As described for food reward, sexual experience can also lead to activation of plasticity-related signaling cascades. The transcription factor delta FosB is increased in the NAc, PFC, dorsal striatum, and VTA following repeated sexual behavior (Wallace et al., 2008; Pitchers et al., 2010b). This natural increase in delta FosB or viral overexpression of delta FosB within the NAc modulates sexual performance, and NAc blockade of delta FosB attenuates this behavior (Hedges et al, 2009; Pitchers et al., 2010b). Further, viral overexpression of delta FosB enhances the conditioned place preference for an environment paired with sexual experience (Hedges et al., 2009). ...
Cite error: The named reference "Natural and drug addictions" was defined multiple times with different content (see the help page).
Table 1 - ^ a b Pitchers KK, Vialou V, Nestler EJ, Laviolette SR, Lehman MN, Coolen LM (February 2013). "Natural and drug rewards act on common neural plasticity mechanisms with ΔFosB as a key mediator". J. Neurosci. 33 (8): 3434–3442. doi:10.1523/JNEUROSCI.4881-12.2013. PMC 3865508. PMID 23426671.
Drugs of abuse induce neuroplasticity in the natural reward pathway, specifically the nucleus accumbens (NAc), thereby causing development and expression of addictive behavior. ... Together, these findings demonstrate that drugs of abuse and natural reward behaviors act on common molecular and cellular mechanisms of plasticity that control vulnerability to drug addiction, and that this increased vulnerability is mediated by ΔFosB and its downstream transcriptional targets. ... Sexual behavior is highly rewarding (Tenk et al., 2009), and sexual experience causes sensitized drug-related behaviors, including cross-sensitization to amphetamine (Amph)-induced locomotor activity (Bradley and Meisel, 2001; Pitchers et al., 2010a) and enhanced Amph reward (Pitchers et al., 2010a). Moreover, sexual experience induces neural plasticity in the NAc similar to that induced by psychostimulant exposure, including increased dendritic spine density (Meisel and Mullins, 2006; Pitchers et al., 2010a), altered glutamate receptor trafficking, and decreased synaptic strength in prefrontal cortex-responding NAc shell neurons (Pitchers et al., 2012). Finally, periods of abstinence from sexual experience were found to be critical for enhanced Amph reward, NAc spinogenesis (Pitchers et al., 2010a), and glutamate receptor trafficking (Pitchers et al., 2012). These findings suggest that natural and drug reward experiences share common mechanisms of neural plasticity
{{cite journal}}: CS1 maint: multiple names: authors list (link) Cite error: The named reference "Amph and sex addiction" was defined multiple times with different content (see the help page). - ^ a b "Entrez Gene: FOSB FBJ murine osteosarcoma viral oncogene homolog B".
- ^ Siderovski DP, Blum S, Forsdyke RE, Forsdyke DR (Oct 1990). "A set of human putative lymphocyte G0/G1 switch genes includes genes homologous to rodent cytokine and zinc finger protein-encoding genes". DNA and Cell Biology. 9 (8): 579–87. doi:10.1089/dna.1990.9.579. PMID 1702972.
- ^ Martin-Gallardo A, McCombie WR, Gocayne JD, FitzGerald MG, Wallace S, Lee BM, Lamerdin J, Trapp S, Kelley JM, Liu LI (Apr 1992). "Automated DNA sequencing and analysis of 106 kilobases from human chromosome 19q13.3". Nature Genetics. 1 (1): 34–9. doi:10.1038/ng0492-34. PMID 1301997. S2CID 1986255.
- ^ Sabatakos G, Rowe GC, Kveiborg M, Wu M, Neff L, Chiusaroli R, Philbrick WM, Baron R (May 2008). "Doubly truncated FosB isoform (Delta2DeltaFosB) induces osteosclerosis in transgenic mice and modulates expression and phosphorylation of Smads in osteoblasts independent of intrinsic AP-1 activity". Journal of Bone and Mineral Research. 23 (5): 584–95. doi:10.1359/jbmr.080110. PMC 2674536. PMID 18433296.
- ^ a b c d e f g h i j k Ruffle JK (Nov 2014). "Molecular neurobiology of addiction: what's all the (Δ)FosB about?". The American Journal of Drug and Alcohol Abuse. 40 (6): 428–37. doi:10.3109/00952990.2014.933840. PMID 25083822. S2CID 19157711.
ΔFosB as a therapeutic biomarker
The strong correlation between chronic drug exposure and ΔFosB provides novel opportunities for targeted therapies in addiction (118), and suggests methods to analyze their efficacy (119). Over the past two decades, research has progressed from identifying ΔFosB induction to investigating its subsequent action (38). It is likely that ΔFosB research will now progress into a new era – the use of ΔFosB as a biomarker. If ΔFosB detection is indicative of chronic drug exposure (and is at least partly responsible for dependence of the substance), then its monitoring for therapeutic efficacy in interventional studies is a suitable biomarker (Figure 2). Examples of therapeutic avenues are discussed herein. ...
Conclusions
ΔFosB is an essential transcription factor implicated in the molecular and behavioral pathways of addiction following repeated drug exposure. The formation of ΔFosB in multiple brain regions, and the molecular pathway leading to the formation of AP-1 complexes is well understood. The establishment of a functional purpose for ΔFosB has allowed further determination as to some of the key aspects of its molecular cascades, involving effectors such as GluR2 (87,88), Cdk5 (93) and NFkB (100). Moreover, many of these molecular changes identified are now directly linked to the structural, physiological and behavioral changes observed following chronic drug exposure (60,95,97,102). New frontiers of research investigating the molecular roles of ΔFosB have been opened by epigenetic studies, and recent advances have illustrated the role of ΔFosB acting on DNA and histones, truly as a molecular switch (34). As a consequence of our improved understanding of ΔFosB in addiction, it is possible to evaluate the addictive potential of current medications (119), as well as use it as a biomarker for assessing the efficacy of therapeutic interventions (121,122,124). Some of these proposed interventions have limitations (125) or are in their infancy (75). However, it is hoped that some of these preliminary findings may lead to innovative treatments, which are much needed in addiction. - ^ a b c d e f g Nestler EJ (December 2013). "Cellular basis of memory for addiction". Dialogues in Clinical Neuroscience. 15 (4): 431–443. PMC 3898681. PMID 24459410.
Despite the importance of numerous psychosocial factors, at its core, drug addiction involves a biological process: the ability of repeated exposure to a drug of abuse to induce changes in a vulnerable brain that drive the compulsive seeking and taking of drugs, and loss of control over drug use, that define a state of addiction. ... A large body of literature has demonstrated that such ΔFosB induction in D1-type [nucleus accumbens] neurons increases an animal's sensitivity to drug as well as natural rewards and promotes drug self-administration, presumably through a process of positive reinforcement ... Another ΔFosB target is cFos: as ΔFosB accumulates with repeated drug exposure it represses c-Fos and contributes to the molecular switch whereby ΔFosB is selectively induced in the chronic drug-treated state.41 ... Moreover, there is increasing evidence that, despite a range of genetic risks for addiction across the population, exposure to sufficiently high doses of a drug for long periods of time can transform someone who has relatively lower genetic loading into an addict.
- ^ a b c Biliński P, Wojtyła A, Kapka-Skrzypczak L, Chwedorowicz R, Cyranka M, Studziński T (2012). "Epigenetic regulation in drug addiction". Annals of Agricultural and Environmental Medicine. 19 (3): 491–6. PMID 23020045.
For these reasons, ΔFosB is considered a primary and causative transcription factor in creating new neural connections in the reward centre, prefrontal cortex, and other regions of the limbic system. This is reflected in the increased, stable and long-lasting level of sensitivity to cocaine and other drugs, and tendency to relapse even after long periods of abstinence. These newly constructed networks function very efficiently via new pathways as soon as drugs of abuse are further taken ... In this way, the induction of CDK5 gene expression occurs together with suppression of the G9A gene coding for dimethyltransferase acting on the histone H3. A feedback mechanism can be observed in the regulation of these 2 crucial factors that determine the adaptive epigenetic response to cocaine. This depends on ΔFosB inhibiting G9a gene expression, i.e. H3K9me2 synthesis which in turn inhibits transcription factors for ΔFosB. For this reason, the observed hyper-expression of G9a, which ensures high levels of the dimethylated form of histone H3, eliminates the neuronal structural and plasticity effects caused by cocaine by means of this feedback which blocks ΔFosB transcription
- ^ Ohnishi YN, Ohnishi YH, Vialou V, Mouzon E, LaPlant Q, Nishi A, Nestler EJ (Jan 2015). "Functional role of the N-terminal domain of ΔFosB in response to stress and drugs of abuse". Neuroscience. 284: 165–70. doi:10.1016/j.neuroscience.2014.10.002. PMC 4268105. PMID 25313003.
- ^ Nakabeppu Y, Nathans D (Feb 1991). "A naturally occurring truncated form of FosB that inhibits Fos/Jun transcriptional activity". Cell. 64 (4): 751–9. doi:10.1016/0092-8674(91)90504-R. PMID 1900040. S2CID 23904956.
- ^ a b c Nestler EJ (Oct 2008). "Review. Transcriptional mechanisms of addiction: role of DeltaFosB". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 363 (1507): 3245–55. doi:10.1098/rstb.2008.0067. PMC 2607320. PMID 18640924.
Recent evidence has shown that ΔFosB also represses the c-fos gene that helps create the molecular switch—from the induction of several short-lived Fos family proteins after acute drug exposure to the predominant accumulation of ΔFosB after chronic drug exposure—cited earlier (Renthal et al. in press). The mechanism responsible for ΔFosB repression of c-fos expression is complex and is covered below. ...
Examples of validated targets for ΔFosB in nucleus accumbens ... GluR2 ... dynorphin ... Cdk5 ... NFκB ... c-Fos
Table 3 - ^ Renthal W, Nestler EJ (Aug 2008). "Epigenetic mechanisms in drug addiction". Trends in Molecular Medicine. 14 (8): 341–50. doi:10.1016/j.molmed.2008.06.004. PMC 2753378. PMID 18635399.
- ^ Renthal W, Kumar A, Xiao G, Wilkinson M, Covington HE, Maze I, Sikder D, Robison AJ, LaPlant Q, Dietz DM, Russo SJ, Vialou V, Chakravarty S, Kodadek TJ, Stack A, Kabbaj M, Nestler EJ (May 2009). "Genome-wide analysis of chromatin regulation by cocaine reveals a role for sirtuins". Neuron. 62 (3): 335–48. doi:10.1016/j.neuron.2009.03.026. PMC 2779727. PMID 19447090.
- ^ Sabatakos G, Sims NA, Chen J, Aoki K, Kelz MB, Amling M, Bouali Y, Mukhopadhyay K, Ford K, Nestler EJ, Baron R (Sep 2000). "Overexpression of DeltaFosB transcription factor(s) increases bone formation and inhibits adipogenesis". Nature Medicine. 6 (9): 985–90. doi:10.1038/79683. PMID 10973317. S2CID 20302360.
- ^ a b c d e Robison AJ, Nestler EJ (November 2011). "Transcriptional and epigenetic mechanisms of addiction". Nature Reviews Neuroscience. 12 (11): 623–637. doi:10.1038/nrn3111. PMC 3272277. PMID 21989194.
ΔFosB serves as one of the master control proteins governing this structural plasticity. ... ΔFosB also represses G9a expression, leading to reduced repressive histone methylation at the cdk5 gene. The net result is gene activation and increased CDK5 expression. ... In contrast, ΔFosB binds to the c-fos gene and recruits several co-repressors, including HDAC1 (histone deacetylase 1) and SIRT 1 (sirtuin 1). ... The net result is c-fos gene repression.
Figure 4: Epigenetic basis of drug regulation of gene expression - ^ a b c d e Nestler EJ, Barrot M, Self DW (Sep 2001). "DeltaFosB: a sustained molecular switch for addiction". Proceedings of the National Academy of Sciences of the United States of America. 98 (20): 11042–6. Bibcode:2001PNAS...9811042N. doi:10.1073/pnas.191352698. PMC 58680. PMID 11572966.
- ^ Salaya-Velazquez NF, López-Muciño LA, Mejía-Chávez S, Sánchez-Aparicio P, Domínguez-Guadarrama AA, Venebra-Muñoz A (February 2020). "Anandamide and sucralose change ΔFosB expression in the reward system". NeuroReport. 31 (3): 240–244. doi:10.1097/WNR.0000000000001400. PMID 31923023. S2CID 210149592.
- ^ Malenka RC, Nestler EJ, Hyman SE (2009). "Chapter 15: Reinforcement and Addictive Disorders". In Sydor A, Brown RY (eds.). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. pp. 364–375. ISBN 978-0-07-148127-4.
- ^ Volkow ND, Koob GF, McLellan AT (January 2016). "Neurobiologic Advances from the Brain Disease Model of Addiction". New England Journal of Medicine. 374 (4): 363–371. doi:10.1056/NEJMra1511480. PMC 6135257. PMID 26816013.
Substance-use disorder: A diagnostic term in the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) referring to recurrent use of alcohol or other drugs that causes clinically and functionally significant impairment, such as health problems, disability, and failure to meet major responsibilities at work, school, or home. Depending on the level of severity, this disorder is classified as mild, moderate, or severe.
Addiction: A term used to indicate the most severe, chronic stage of substance-use disorder, in which there is a substantial loss of self-control, as indicated by compulsive drug taking despite the desire to stop taking the drug. In the DSM-5, the term addiction is synonymous with the classification of severe substance-use disorder. - ^ a b c Renthal W, Nestler EJ (September 2009). "Chromatin regulation in drug addiction and depression". Dialogues in Clinical Neuroscience. 11 (3): 257–268. doi:10.31887/DCNS.2009.11.3/wrenthal. PMC 2834246. PMID 19877494.
[Psychostimulants] increase cAMP levels in striatum, which activates protein kinase A (PKA) and leads to phosphorylation of its targets. This includes the cAMP response element binding protein (CREB), the phosphorylation of which induces its association with the histone acetyltransferase, CREB binding protein (CBP) to acetylate histones and facilitate gene activation. This is known to occur on many genes including fosB and c-fos in response to psychostimulant exposure. ΔFosB is also upregulated by chronic psychostimulant treatments, and is known to activate certain genes (eg, cdk5) and repress others (eg, c-fos) where it recruits HDAC1 as a corepressor. ... Chronic exposure to psychostimulants increases glutamatergic [signaling] from the prefrontal cortex to the NAc. Glutamatergic signaling elevates Ca2+ levels in NAc postsynaptic elements where it activates CaMK (calcium/calmodulin protein kinases) signaling, which, in addition to phosphorylating CREB, also phosphorylates HDAC5.
Figure 2: Psychostimulant-induced signaling events - ^ Broussard JI (January 2012). "Co-transmission of dopamine and glutamate". The Journal of General Physiology. 139 (1): 93–96. doi:10.1085/jgp.201110659. PMC 3250102. PMID 22200950.
Coincident and convergent input often induces plasticity on a postsynaptic neuron. The NAc integrates processed information about the environment from basolateral amygdala, hippocampus, and prefrontal cortex (PFC), as well as projections from midbrain dopamine neurons. Previous studies have demonstrated how dopamine modulates this integrative process. For example, high frequency stimulation potentiates hippocampal inputs to the NAc while simultaneously depressing PFC synapses (Goto and Grace, 2005). The converse was also shown to be true; stimulation at PFC potentiates PFC–NAc synapses but depresses hippocampal–NAc synapses. In light of the new functional evidence of midbrain dopamine/glutamate co-transmission (references above), new experiments of NAc function will have to test whether midbrain glutamatergic inputs bias or filter either limbic or cortical inputs to guide goal-directed behavior.
- ^ Kanehisa Laboratories (10 October 2014). "Amphetamine – Homo sapiens (human)". KEGG Pathway. Retrieved 31 October 2014.
Most addictive drugs increase extracellular concentrations of dopamine (DA) in nucleus accumbens (NAc) and medial prefrontal cortex (mPFC), projection areas of mesocorticolimbic DA neurons and key components of the "brain reward circuit". Amphetamine achieves this elevation in extracellular levels of DA by promoting efflux from synaptic terminals. ... Chronic exposure to amphetamine induces a unique transcription factor delta FosB, which plays an essential role in long-term adaptive changes in the brain.
- ^ Cadet JL, Brannock C, Jayanthi S, Krasnova IN (2015). "Transcriptional and epigenetic substrates of methamphetamine addiction and withdrawal: evidence from a long-access self-administration model in the rat". Molecular Neurobiology. 51 (2): 696–717 (Figure 1). doi:10.1007/s12035-014-8776-8. PMC 4359351. PMID 24939695.
- ^ a b c d Nestler EJ (December 2012). "Transcriptional mechanisms of drug addiction". Clinical Psychopharmacology and Neuroscience. 10 (3): 136–143. doi:10.9758/cpn.2012.10.3.136. PMC 3569166. PMID 23430970.
The 35-37 kD ΔFosB isoforms accumulate with chronic drug exposure due to their extraordinarily long half-lives. ... As a result of its stability, the ΔFosB protein persists in neurons for at least several weeks after cessation of drug exposure. ... ΔFosB overexpression in nucleus accumbens induces NFκB ... In contrast, the ability of ΔFosB to repress the c-Fos gene occurs in concert with the recruitment of a histone deacetylase and presumably several other repressive proteins such as a repressive histone methyltransferase
- ^ Nestler EJ (October 2008). "Transcriptional mechanisms of addiction: Role of ΔFosB". Philosophical Transactions of the Royal Society B: Biological Sciences. 363 (1507): 3245–3255. doi:10.1098/rstb.2008.0067. PMC 2607320. PMID 18640924.
Recent evidence has shown that ΔFosB also represses the c-fos gene that helps create the molecular switch—from the induction of several short-lived Fos family proteins after acute drug exposure to the predominant accumulation of ΔFosB after chronic drug exposure
- ^ Kim Y, Teylan MA, Baron M, Sands A, Nairn AC, Greengard P (Feb 2009). "Methylphenidate-induced dendritic spine formation and DeltaFosB expression in nucleus accumbens". Proceedings of the National Academy of Sciences of the United States of America. 106 (8): 2915–20. Bibcode:2009PNAS..106.2915K. doi:10.1073/pnas.0813179106. PMC 2650365. PMID 19202072.
- ^ a b c d Nestler EJ (January 2014). "Epigenetic mechanisms of drug addiction". Neuropharmacology. 76 Pt B: 259–268. doi:10.1016/j.neuropharm.2013.04.004. PMC 3766384. PMID 23643695.
Short-term increases in histone acetylation generally promote behavioral responses to the drugs, while sustained increases oppose cocaine's effects, based on the actions of systemic or intra-NAc administration of HDAC inhibitors. ... Genetic or pharmacological blockade of G9a in the NAc potentiates behavioral responses to cocaine and opiates, whereas increasing G9a function exerts the opposite effect (Maze et al., 2010; Sun et al., 2012a). Such drug-induced downregulation of G9a and H3K9me2 also sensitizes animals to the deleterious effects of subsequent chronic stress (Covington et al., 2011). Downregulation of G9a increases the dendritic arborization of NAc neurons, and is associated with increased expression of numerous proteins implicated in synaptic function, which directly connects altered G9a/H3K9me2 in the synaptic plasticity associated with addiction (Maze et al., 2010).
G9a appears to be a critical control point for epigenetic regulation in NAc, as we know it functions in two negative feedback loops. It opposes the induction of ΔFosB, a long-lasting transcription factor important for drug addiction (Robison and Nestler, 2011), while ΔFosB in turn suppresses G9a expression (Maze et al., 2010; Sun et al., 2012a). ... Also, G9a is induced in NAc upon prolonged HDAC inhibition, which explains the paradoxical attenuation of cocaine's behavioral effects seen under these conditions, as noted above (Kennedy et al., 2013). GABAA receptor subunit genes are among those that are controlled by this feedback loop. Thus, chronic cocaine, or prolonged HDAC inhibition, induces several GABAA receptor subunits in NAc, which is associated with increased frequency of inhibitory postsynaptic currents (IPSCs). In striking contrast, combined exposure to cocaine and HDAC inhibition, which triggers the induction of G9a and increased global levels of H3K9me2, leads to blockade of GABAA receptor and IPSC regulation. - ^ Malenka RC, Nestler EJ, Hyman SE (2009). "Chapter 15: Reinforcement and addictive disorders". In Sydor A, Brown RY (eds.). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. pp. 384–385. ISBN 9780071481274.
- ^ a b McCowan TJ, Dhasarathy A, Carvelli L (February 2015). "The Epigenetic Mechanisms of Amphetamine". J. Addict. Prev. 2015 (Suppl 1). PMC 4955852. PMID 27453897.
Epigenetic modifications caused by addictive drugs play an important role in neuronal plasticity and in drug-induced behavioral responses. Although few studies have investigated the effects of AMPH on gene regulation (Table 1), current data suggest that AMPH acts at multiple levels to alter histone/DNA interaction and to recruit transcription factors which ultimately cause repression of some genes and activation of other genes. Importantly, some studies have also correlated the epigenetic regulation induced by AMPH with the behavioral outcomes caused by this drug, suggesting therefore that epigenetics remodeling underlies the behavioral changes induced by AMPH. If this proves to be true, the use of specific drugs that inhibit histone acetylation, methylation or DNA methylation might be an important therapeutic alternative to prevent and/or reverse AMPH addiction and mitigate the side effects generate by AMPH when used to treat ADHD.
- ^ a b c d Walker DM, Cates HM, Heller EA, Nestler EJ (February 2015). "Regulation of chromatin states by drugs of abuse". Curr. Opin. Neurobiol. 30: 112–121. doi:10.1016/j.conb.2014.11.002. PMC 4293340. PMID 25486626.
Studies investigating general HDAC inhibition on behavioral outcomes have produced varying results but it seems that the effects are specific to the timing of exposure (either before, during or after exposure to drugs of abuse) as well as the length of exposure
- ^ a b Primary references involving sodium butyrate:
• Kennedy PJ, Feng J, Robison AJ, Maze I, Badimon A, Mouzon E, Chaudhury D, Damez-Werno DM, Haggarty SJ, Han MH, Bassel-Duby R, Olson EN, Nestler EJ (April 2013). "Class I HDAC inhibition blocks cocaine-induced plasticity by targeted changes in histone methylation". Nat. Neurosci. 16 (4): 434–440. doi:10.1038/nn.3354. PMC 3609040. PMID 23475113.While acute HDAC inhibition enhances the behavioral effects of cocaine or amphetamine1,3,4,13,14, studies suggest that more chronic regimens block psychostimulant-induced plasticity3,5,11,12. ... The effects of pharmacological inhibition of HDACs on psychostimulant-induced plasticity appear to depend on the timecourse of HDAC inhibition. Studies employing co-administration procedures in which inhibitors are given acutely, just prior to psychostimulant administration, report heightened behavioral responses to the drug1,3,4,13,14. In contrast, experimental paradigms like the one employed here, in which HDAC inhibitors are administered more chronically, for several days prior to psychostimulant exposure, show inhibited expression3 or decreased acquisition of behavioral adaptations to drug5,11,12. The clustering of seemingly discrepant results based on experimental methodologies is interesting in light of our present findings. Both HDAC inhibitors and psychostimulants increase global levels of histone acetylation in NAc. Thus, when co-administered acutely, these drugs may have synergistic effects, leading to heightened transcriptional activation of psychostimulant-regulated target genes. In contrast, when a psychostimulant is given in the context of prolonged, HDAC inhibitor-induced hyperacetylation, homeostatic processes may direct AcH3 binding to the promoters of genes (e.g., G9a) responsible for inducing chromatin condensation and gene repression (e.g., via H3K9me2) in order to dampen already heightened transcriptional activation. Our present findings thus demonstrate clear cross talk among histone PTMs and suggest that decreased behavioral sensitivity to psychostimulants following prolonged HDAC inhibition might be mediated through decreased activity of HDAC1 at H3K9 KMT promoters and subsequent increases in H3K9me2 and gene repression.
• Simon-O'Brien E, Alaux-Cantin S, Warnault V, Buttolo R, Naassila M, Vilpoux C (July 2015). "The histone deacetylase inhibitor sodium butyrate decreases excessive ethanol intake in dependent animals". Addict Biol. 20 (4): 676–689. doi:10.1111/adb.12161. PMID 25041570. S2CID 28667144.Altogether, our results clearly demonstrated the efficacy of NaB in preventing excessive ethanol intake and relapse and support the hypothesis that HDACi may have a potential use in alcohol addiction treatment.
• Castino MR, Cornish JL, Clemens KJ (April 2015). "Inhibition of histone deacetylases facilitates extinction and attenuates reinstatement of nicotine self-administration in rats". PLOS ONE. 10 (4): e0124796. Bibcode:2015PLoSO..1024796C. doi:10.1371/journal.pone.0124796. PMC 4399837. PMID 25880762.treatment with NaB significantly attenuated nicotine and nicotine + cue reinstatement when administered immediately ... These results provide the first demonstration that HDAC inhibition facilitates the extinction of responding for an intravenously self-administered drug of abuse and further highlight the potential of HDAC inhibitors in the treatment of drug addiction.
- ^ Kyzar EJ, Pandey SC (August 2015). "Molecular mechanisms of synaptic remodeling in alcoholism". Neurosci. Lett. 601: 11–9. doi:10.1016/j.neulet.2015.01.051. PMC 4506731. PMID 25623036.
Increased HDAC2 expression decreases the expression of genes important for the maintenance of dendritic spine density such as BDNF, Arc, and NPY, leading to increased anxiety and alcohol-seeking behavior. Decreasing HDAC2 reverses both the molecular and behavioral consequences of alcohol addiction, thus implicating this enzyme as a potential treatment target (Fig. 3). HDAC2 is also crucial for the induction and maintenance of structural synaptic plasticity in other neurological domains such as memory formation [115]. Taken together, these findings underscore the potential usefulness of HDAC inhibition in treating alcohol use disorders ... Given the ability of HDAC inhibitors to potently modulate the synaptic plasticity of learning and memory [118], these drugs hold potential as treatment for substance abuse-related disorders. ... Our lab and others have published extensively on the ability of HDAC inhibitors to reverse the gene expression deficits caused by multiple models of alcoholism and alcohol abuse, the results of which were discussed above [25,112,113]. This data supports further examination of histone modifying agents as potential therapeutic drugs in the treatment of alcohol addiction ... Future studies should continue to elucidate the specific epigenetic mechanisms underlying compulsive alcohol use and alcoholism, as this is likely to provide new molecular targets for clinical intervention.
- ^ Kheirabadi GR, Ghavami M, Maracy MR, Salehi M, Sharbafchi MR (2016). "Effect of add-on valproate on craving in methamphetamine depended patients: A randomized trial". Advanced Biomedical Research. 5: 149. doi:10.4103/2277-9175.187404. PMC 5025910. PMID 27656618.
- ^ Hope BT (May 1998). "Cocaine and the AP-1 transcription factor complex". Annals of the New York Academy of Sciences. 844 (1): 1–6. Bibcode:1998NYASA.844....1H. doi:10.1111/j.1749-6632.1998.tb08216.x. PMID 9668659. S2CID 11683570.
- ^ a b Kelz MB, Chen J, Carlezon WA, Whisler K, Gilden L, Beckmann AM, Steffen C, Zhang YJ, Marotti L, Self DW, Tkatch T, Baranauskas G, Surmeier DJ, Neve RL, Duman RS, Picciotto MR, Nestler EJ (Sep 1999). "Expression of the transcription factor deltaFosB in the brain controls sensitivity to cocaine". Nature. 401 (6750): 272–6. Bibcode:1999Natur.401..272K. doi:10.1038/45790. PMID 10499584. S2CID 4390717.
- ^ a b Colby CR, Whisler K, Steffen C, Nestler EJ, Self DW (Mar 2003). "Striatal cell type-specific overexpression of DeltaFosB enhances incentive for cocaine". The Journal of Neuroscience. 23 (6): 2488–93. doi:10.1523/JNEUROSCI.23-06-02488.2003. PMC 6742034. PMID 12657709.
- ^ Cao X, Yasuda T, Uthayathas S, Watts RL, Mouradian MM, Mochizuki H, Papa SM (May 2010). "Striatal overexpression of DeltaFosB reproduces chronic levodopa-induced involuntary movements". The Journal of Neuroscience. 30 (21): 7335–43. doi:10.1523/JNEUROSCI.0252-10.2010. PMC 2888489. PMID 20505100.
- ^ a b c d e Du H, Nie S, Chen G, Ma K, Xu Y, Zhang Z, Papa SM, Cao X (2015). "Levetiracetam Ameliorates L-DOPA-Induced Dyskinesia in Hemiparkinsonian Rats Inducing Critical Molecular Changes in the Striatum". Parkinson's Disease. 2015: 253878. doi:10.1155/2015/253878. PMC 4322303. PMID 25692070.
Furthermore, the transgenic overexpression of ΔFosB reproduces AIMs in hemiparkinsonian rats without chronic exposure to L-DOPA [13]. ... FosB/ΔFosB immunoreactive neurons increased in the dorsolateral part of the striatum on the lesion side with the used antibody that recognizes all members of the FosB family. All doses of levetiracetam decreased the number of FosB/ΔFosB positive cells (from 88.7 ± 1.7/section in the control group to 65.7 ± 0.87, 42.3 ± 1.88, and 25.7 ± 1.2/section in the 15, 30, and 60 mg groups, resp.; Figure 2). These results indicate dose-dependent effects of levetiracetam on FosB/ΔFosB expression. ... In addition, transcription factors expressed with chronic events such as ΔFosB (a truncated splice variant of FosB) are overexpressed in the striatum of rodents and primates with dyskinesias [9, 10]. ... Furthermore, ΔFosB overexpression has been observed in postmortem striatal studies of Parkinsonian patients chronically treated with L-DOPA [26]. ... Of note, the most prominent effect of levetiracetam was the reduction of ΔFosB expression, which cannot be explained by any of its known actions on vesicular protein or ion channels. Therefore, the exact mechanism(s) underlying the antiepileptic effects of levetiracetam remains uncertain.
- ^ "ROLE OF ΔFOSB IN THE NUCLEUS ACCUMBENS". Mount Sinai School of Medicine. NESTLER LAB: LABORATORY OF MOLECULAR PSYCHIATRY. Archived from the original on 28 June 2017. Retrieved 6 September 2014.
- ^ Furuyashiki T, Deguchi Y (Aug 2012). "[Roles of altered striatal function in major depression]". Brain and Nerve = Shinkei Kenkyū No Shinpo (in Japanese). 64 (8): 919–26. PMID 22868883.
- ^ Nestler EJ (Apr 2015). "∆FosB: a transcriptional regulator of stress and antidepressant responses". European Journal of Pharmacology. 753: 66–72. doi:10.1016/j.ejphar.2014.10.034. PMC 4380559. PMID 25446562.
In more recent years, prolonged induction of ∆FosB has also been observed within NAc in response to chronic administration of certain forms of stress. Increasing evidence indicates that this induction represents a positive, homeostatic adaptation to chronic stress, since overexpression of ∆FosB in this brain region promotes resilience to stress, whereas blockade of its activity promotes stress susceptibility. Chronic administration of several antidepressant medications also induces ∆FosB in the NAc, and this induction is required for the therapeutic-like actions of these drugs in mouse models. Validation of these rodent findings is the demonstration that depressed humans, examined at autopsy, display reduced levels of ∆FosB within the NAc. As a transcription factor, ΔFosB produces this behavioral phenotype by regulating the expression of specific target genes, which are under current investigation. These studies of ΔFosB are providing new insight into the molecular basis of depression and antidepressant action, which is defining a host of new targets for possible therapeutic development.
- ^ Dietz DM, Kennedy PJ, Sun H, Maze I, Gancarz AM, Vialou V, Koo JW, Mouzon E, Ghose S, Tamminga CA, Nestler EJ (February 2014). "ΔFosB induction in prefrontal cortex by antipsychotic drugs is associated with negative behavioral outcomes". Neuropsychopharmacology. 39 (3): 538–44. doi:10.1038/npp.2013.255. PMC 3895248. PMID 24067299.
Further reading
- Schuermann M, Jooss K, Müller R (Apr 1991). "fosB is a transforming gene encoding a transcriptional activator". Oncogene. 6 (4): 567–76. PMID 1903195.
- Brown JR, Ye H, Bronson RT, Dikkes P, Greenberg ME (Jul 1996). "A defect in nurturing in mice lacking the immediate early gene fosB". Cell. 86 (2): 297–309. doi:10.1016/S0092-8674(00)80101-4. PMID 8706134. S2CID 17266171.
- Heximer SP, Cristillo AD, Russell L, Forsdyke DR (Dec 1996). "Sequence analysis and expression in cultured lymphocytes of the human FOSB gene (G0S3)". DNA and Cell Biology. 15 (12): 1025–38. doi:10.1089/dna.1996.15.1025. PMID 8985116.
- Liberati NT, Datto MB, Frederick JP, Shen X, Wong C, Rougier-Chapman EM, Wang XF (Apr 1999). "Smads bind directly to the Jun family of AP-1 transcription factors". Proceedings of the National Academy of Sciences of the United States of America. 96 (9): 4844–9. Bibcode:1999PNAS...96.4844L. doi:10.1073/pnas.96.9.4844. PMC 21779. PMID 10220381.
- Yamamura Y, Hua X, Bergelson S, Lodish HF (Nov 2000). "Critical role of Smads and AP-1 complex in transforming growth factor-beta -dependent apoptosis". The Journal of Biological Chemistry. 275 (46): 36295–302. doi:10.1074/jbc.M006023200. PMID 10942775.
- Bergman MR, Cheng S, Honbo N, Piacentini L, Karliner JS, Lovett DH (Feb 2003). "A functional activating protein 1 (AP-1) site regulates matrix metalloproteinase 2 (MMP-2) transcription by cardiac cells through interactions with JunB-Fra1 and JunB-FosB heterodimers". Biochemical Journal. 369 (Pt 3): 485–96. doi:10.1042/BJ20020707. PMC 1223099. PMID 12371906.
- Milde-Langosch K, Kappes H, Riethdorf S, Löning T, Bamberger AM (Feb 2003). "FosB is highly expressed in normal mammary epithelia, but down-regulated in poorly differentiated breast carcinomas". Breast Cancer Research and Treatment. 77 (3): 265–75. doi:10.1023/A:1021887100216. PMID 12602926. S2CID 987857.
- Baumann S, Hess J, Eichhorst ST, Krueger A, Angel P, Krammer PH, Kirchhoff S (Mar 2003). "An unexpected role for FosB in activation-induced cell death of T cells". Oncogene. 22 (9): 1333–9. doi:10.1038/sj.onc.1206126. PMID 12618758. S2CID 10696422.
- Holmes DI, Zachary I (Jan 2004). "Placental growth factor induces FosB and c-Fos gene expression via Flt-1 receptors". FEBS Letters. 557 (1–3): 93–8. Bibcode:2004FEBSL.557...93H. doi:10.1016/S0014-5793(03)01452-2. PMID 14741347. S2CID 6596900.
- Konsman JP, Blomqvist A (May 2005). "Forebrain patterns of c-Fos and FosB induction during cancer-associated anorexia-cachexia in rat". The European Journal of Neuroscience. 21 (10): 2752–66. doi:10.1111/j.1460-9568.2005.04102.x. PMID 15926923. S2CID 40045788.
External links
- ROLE OF ΔFOSB IN THE NUCLEUS ACCUMBENS Archived 28 June 2017 at the Wayback Machine
- KEGG Pathway – human alcohol addiction
- KEGG Pathway – human amphetamine addiction
- KEGG Pathway – human cocaine addiction
- FOSB+protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
This article incorporates text from the United States National Library of Medicine, which is in the public domain.
Depression
In April 2007, two research teams reported on having inserted electrodes into the nucleus accumbens in order to use deep brain stimulation to treat severe depression.[1] In 2010 experiments reported that deep brain stimulation of the nucleus accumbens was successful in decreasing depression symptoms in 50% of patients who did not respond to other treatments such as electroconvulsive therapy.[2] Nucleus accumbens has also been used as a target to treat small groups of patients with therapy-refractory obsessive-compulsive disorder.[3]
Placebo effect
Activation of the NAcc has been shown to occur in the anticipation of effectiveness of a drug when a user is given a placebo, indicating a contributing role of the nucleus accumbens in the placebo effect.[4][5]
Additional images
-
Dopamine and serotonin
-
MRI coronal slice showing nucleus accumbens outlined in red
-
Sagittal MRI slice with highlighting (red) indicating the nucleus accumbens.
References
- ^ Brain Electrodes Help Treat Depression, Technology Review, 26 April 2007
- ^ Bewernick, B. H., Hurlemann, R., Matusch, A., et al. Nucleus accumbens deep brain stimulation decreases ratings of depression and anciety in treatment-resistant depression. Biological Psychiatry', 2009 67: 110-116.
- ^ Ooms P, Mantione M, Figee M, Schuurman PR, van den Munckhof P, Denys D. Deep brain stimulation for obsessive-compulsive disorders: long-term analysis of quality of life. J Neurol Neurosurg Psychiatry. 2014;85(2):153-8.
- ^ http://www.eurekalert.org/pub_releases/2007-07/cp-brc071607.php Brain region central to placebo effect identified
- ^ Cite error: The named reference
placebo effectwas invoked but never defined (see the help page).
External links
- The role of the nucleus accumbens in the reward circuit. Part of "The Brain From Top to Bottom." at thebrain.mcgill.ca
- Nucleus Accumbens - Cell Centered Database
- Stained brain slice images which include the "nucleus%20accumbens" at the BrainMaps project