Search results
Appearance
There is a page named "CaC2O4" on Wikipedia
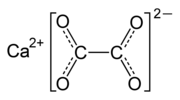
- Calcium oxalate (redirect from CaC2O4)calcium salt of oxalic acid with the chemical formula CaC2O4 or Ca(COO)2. It forms hydrates CaC2O4·nH2O, where n varies from 1 to 3. Anhydrous and all hydrated...16 KB (1,485 words) - 06:02, 27 June 2024
- resulting reaction is as follows: CaCO3(s) + H2C2O4(l) → CaC2O4(s) + CO2(g) + H2O(l) In this case the calcium oxalate (CaC2O4) formed in the reaction is washed...36 KB (3,216 words) - 11:41, 13 June 2024
- Calcium hypochlorite (redirect from Ca(ClO)2)hypochlorite is an inorganic compound with chemical formula Ca(ClO)2, also written as Ca(OCl)2. It is a white solid, although commercial samples appear...9 KB (822 words) - 07:10, 2 April 2024
- Calcium carbide (redirect from CaC2)as calcium acetylide, is a chemical compound with the chemical formula of CaC2. Its main use industrially is in the production of acetylene and calcium...17 KB (1,660 words) - 16:20, 3 June 2024
- entirely to calcium oxide. The reaction is pure calcium oxide formed CaC2O4 → CaO(s) + CO(g)+ CO2(g) The pure precipitate is cooled, then measured by...22 KB (2,986 words) - 19:03, 30 March 2023
- Calcium nitrate (redirect from Ca(NO3)2.4H2O)Calcium nitrate are inorganic compounds with the formula Ca(NO3)2(H2O)x. The anhydrous compound, which is rarely encountered, absorbs moisture from the...12 KB (1,038 words) - 08:54, 12 June 2024
- Calcium sulfate (redirect from CaSO4)sulfate (or calcium sulphate) is the inorganic compound with the formula CaSO4 and related hydrates. In the form of γ-anhydrite (the anhydrous form),...19 KB (1,828 words) - 19:45, 7 June 2024
- Calcium hydroxide (redirect from Ca(OH)2)(traditionally called slaked lime) is an inorganic compound with the chemical formula Ca(OH)2. It is a colorless crystal or white powder and is produced when quicklime...22 KB (2,117 words) - 20:00, 26 June 2024
- calcium oxalate monohydrate, or whewellite (CaC2O4·H2O), and calcium oxalate dihydrate, or weddellite (CaC2O4·2H2O). Their appearance can be rough, smooth...18 KB (2,216 words) - 11:47, 15 July 2024
- Calcium carbonate (redirect from CaCO3)Calcium carbonate is a chemical compound with the chemical formula CaCO3. It is a common substance found in rocks as the minerals calcite and aragonite...79 KB (7,564 words) - 14:25, 12 July 2024
- Monocalcium phosphate (redirect from Ca(H2PO4)2)phosphoric acid: Ca(OH)2 + 2 H3PO4 → Ca(H2PO4)2 + 2 H2O Samples of Ca(H2PO4)2 tend to convert to dicalcium phosphate: Ca(H2PO4)2 → Ca(HPO4) + H3PO4 Superphosphate...7 KB (524 words) - 09:30, 11 June 2024
- Calcium fluoride (redirect from CaF2)inorganic compound of the elements calcium and fluorine with the formula CaF2. It is a white solid that is practically insoluble in water. It occurs as...10 KB (843 words) - 22:46, 14 January 2024
- Weddellite (CaC2O4·2H2O) is a mineral form of calcium oxalate named for occurrences of millimeter-sized crystals found in bottom sediments of the Weddell...4 KB (256 words) - 06:40, 11 May 2023
- chemical compound which is a calcium salt of acetic acid. It has the formula Ca(C2H3O2)2. Its standard name is calcium acetate, while calcium ethanoate is...8 KB (531 words) - 23:45, 3 March 2024
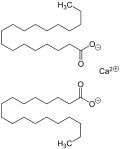
- stearate is produced by heating stearic acid and calcium oxide: 2 C17H35COOH + CaO → (C17H35COO)2Ca + H2O It is also the main component of soap scum, a white...5 KB (458 words) - 00:59, 8 March 2023
- employed, involving a soluble phosphate and calcium salts, e.g. (NH4)2HPO4 + Ca(NO3)2. is performed under carefully controlled pH conditions. The precipitate...18 KB (1,828 words) - 20:19, 24 April 2024
- Ca(H2PO4)2 and Ca(H2PO4)2(H2O) Dicalcium phosphate (dibasic calcium phosphate), E341(ii) (CAS# 7757-93-9): CaHPO4 (mineral: monetite), dihydrate CaHPO4(H2O)2...7 KB (602 words) - 03:25, 18 May 2024
- (chemical formula CaTiO3). Its name is also applied to the class of compounds which have the same type of crystal structure as CaTiO3, known as the perovskite...16 KB (1,588 words) - 08:04, 26 April 2024
- Calcium oxide (redirect from CaO)Calcium oxide (formula: CaO), commonly known as quicklime or burnt lime, is a widely used chemical compound. It is a white, caustic, alkaline, crystalline...23 KB (2,231 words) - 06:34, 23 June 2024
- 2 O 4 + CaCl 2 = 2 NaCl + CaC 2 O 4 {\displaystyle {\ce {Na2C2O4 + CaCl2}}={\ce {2NaCl + CaC2O4}}} . Na 2 C 2 O 4 + CaSO 4 = Na 2 SO 4 + CaC 2 O 4 {\displaystyle
- 2MnO4- + 16H+ + 5C2O4-2 ---------> 2Mn+2 +8H2O + 10 CO2 Oxalate ions are C2O4-- Concentrated sulphuric acid decomposes oxalate ions into carbon monoxide