Calcium sulfate

| |

| |
| Names | |
|---|---|
| Other names | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI |
|
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.029.000 |
| EC Number |
|
| E number | E516 (acidity regulators, ...) |
| 7487 | |
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CaSO4 | |
| Molar mass | 136.14 g/mol (anhydrous) 145.15 g/mol (hemihydrate) 172.172 g/mol (dihydrate) |
| Appearance | white solid |
| Odor | odorless |
| Density | 2.96 g/cm3 (anhydrous) 2.32 g/cm3 (dihydrate) |
| Melting point | 1,460 °C (2,660 °F; 1,730 K) (anhydrous) |
| 0.26 g/100ml at 25 °C (dihydrate)[1] | |
Solubility product (Ksp)
|
4.93 × 10−5 mol2L−2 (anhydrous) 3.14 × 10−5 (dihydrate) [2] |
| Solubility in glycerol | slightly soluble (dihydrate) |
| Acidity (pKa) | 10.4 (anhydrous) 7.3 (dihydrate) |
| -49.7·10−6 cm3/mol | |
| Structure | |
| orthorhombic | |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
107 J·mol−1·K−1 [3] |
Std enthalpy of
formation (ΔfH⦵298) |
-1433 kJ/mol[3] |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 15 mg/m3 (total) TWA 5 mg/m3 (resp) [for anhydrous form only][4] |
REL (Recommended)
|
TWA 10 mg/m3 (total) TWA 5 mg/m3 (resp) [anhydrous only][4] |
IDLH (Immediate danger)
|
N.D.[4] |
| Safety data sheet (SDS) | ICSC 1589 |
| Related compounds | |
Other cations
|
Magnesium sulfate Strontium sulfate Barium sulfate |
Related desiccants
|
Calcium chloride Magnesium sulfate |
Related compounds
|
Plaster of Paris Gypsum |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Calcium sulfate (or calcium sulphate) is the inorganic compound with the formula CaSO4 and related hydrates. In the form of γ-anhydrite (the anhydrous form), it is used as a desiccant. One particular hydrate is better known as plaster of Paris, and another occurs naturally as the mineral gypsum. It has many uses in industry. All forms are white solids that are poorly soluble in water.[5] Calcium sulfate causes permanent hardness in water.
Hydration states and crystallographic structures
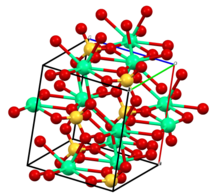
The compound exists in three levels of hydration corresponding to different crystallographic structures and to minerals:
- CaSO
4 (anhydrite): anhydrous state.[6] The structure is related to that of zirconium orthosilicate (zircon): Ca2+
is 8-coordinate, SO2−
4 is tetrahedral, O is 3-coordinate. - CaSO
4·2H
2O (gypsum and selenite (mineral)): dihydrate.[7] - CaSO
4·1/2H
2O (bassanite): hemihydrate, also known as plaster of Paris. Specific hemihydrates are sometimes distinguished: α-hemihydrate and β-hemihydrate.[8]
Uses
The main use of calcium sulfate is to produce plaster of Paris and stucco. These applications exploit the fact that calcium sulfate which has been powdered and calcined forms a moldable paste upon hydration and hardens as crystalline calcium sulfate dihydrate. It is also convenient that calcium sulfate is poorly soluble in water and does not readily dissolve in contact with water after its solidification.
Hydration and dehydration reactions
With judicious heating, gypsum converts to the partially dehydrated mineral called bassanite or plaster of Paris. This material has the formula CaSO4·(nH2O), where 0.5 ≤ n ≤ 0.8.[8] Temperatures between 100 and 150 °C (212–302 °F) are required to drive off the water within its structure. The details of the temperature and time depend on ambient humidity. Temperatures as high as 170 °C (338 °F) are used in industrial calcination, but at these temperatures γ-anhydrite begins to form. The heat energy delivered to the gypsum at this time (the heat of hydration) tends to go into driving off water (as water vapor) rather than increasing the temperature of the mineral, which rises slowly until the water is gone, then increases more rapidly. The equation for the partial dehydration is:
- CaSO4 · 2 H2O → CaSO4 · 1/2 H2O + 1+1/2 H2O↑
The endothermic property of this reaction is relevant to the performance of drywall, conferring fire resistance to residential and other structures. In a fire, the structure behind a sheet of drywall will remain relatively cool as water is lost from the gypsum, thus preventing (or substantially retarding) damage to the framing (through combustion of wood members or loss of strength of steel at high temperatures) and consequent structural collapse. But at higher temperatures, calcium sulfate will release oxygen and act as an oxidizing agent. This property is used in aluminothermy. In contrast to most minerals, which when rehydrated simply form liquid or semi-liquid pastes, or remain powdery, calcined gypsum has an unusual property: when mixed with water at normal (ambient) temperatures, it quickly reverts chemically to the preferred dihydrate form, while physically "setting" to form a rigid and relatively strong gypsum crystal lattice:
- CaSO4 · 1/2 H2O + 1+1/2 H2O → CaSO4 · 2 H2O
This reaction is exothermic and is responsible for the ease with which gypsum can be cast into various shapes including sheets (for drywall), sticks (for blackboard chalk), and molds (to immobilize broken bones, or for metal casting). Mixed with polymers, it has been used as a bone repair cement. Small amounts of calcined gypsum are added to earth to create strong structures directly from cast earth, an alternative to adobe (which loses its strength when wet). The conditions of dehydration can be changed to adjust the porosity of the hemihydrate, resulting in the so-called α- and β-hemihydrates (which are more or less chemically identical).
On heating to 180 °C (356 °F), the nearly water-free form, called γ-anhydrite (CaSO4·nH2O where n = 0 to 0.05) is produced. γ-Anhydrite reacts slowly with water to return to the dihydrate state, a property exploited in some commercial desiccants. On heating above 250 °C, the completely anhydrous form called β-anhydrite or "natural" anhydrite is formed. Natural anhydrite does not react with water, even over geological timescales, unless very finely ground.
The variable composition of the hemihydrate and γ-anhydrite, and their easy inter-conversion, is due to their nearly identical crystal structures containing "channels" that can accommodate variable amounts of water, or other small molecules such as methanol.
Food industry
The calcium sulfate hydrates are used as a coagulant in products such as tofu.[9]
For the FDA, it is permitted in cheese and related cheese products; cereal flours; bakery products; frozen desserts; artificial sweeteners for jelly & preserves; condiment vegetables; and condiment tomatoes and some candies.[10]
It is known in the E number series as E516, and the UN's FAO knows it as a firming agent, a flour treatment agent, a sequestrant, and a leavening agent.[10]
Dentistry
Calcium sulfate has a long history of use in dentistry.[11] It has been used in bone regeneration as a graft material and graft binder (or extender) and as a barrier in guided bone tissue regeneration. It is a biocompatible material and is completely resorbed following implantation.[12] It does not evoke a significant host response and creates a calcium-rich milieu in the area of implantation.[13]
Desiccant

When sold at the anhydrous state as a desiccant with a color-indicating agent under the name Drierite, it appears blue (anhydrous) or pink (hydrated) due to impregnation with cobalt(II) chloride, which functions as a moisture indicator.
Sulfuric acid production
Up to the 1970s, commercial quantities of sulfuric acid were produced from anhydrous calcium sulfate.[14] Upon being mixed with shale or marl, and roasted at 1400°C, the sulfate liberates sulfur dioxide gas, a precursor to sulfuric acid. The reaction also produces calcium silicate, used in cement clinker production.[15][16]
- 2 CaSO4 + 2 SiO2 + C → 2 CaSiO3 + 2 SO2 + CO2
Some component reactions pertaining to calcium sulfate:
- CaSO4 + 2 C → CaS + 2 CO2
- 3 CaSO4 + CaS + 2 SiO2 → 2 Ca2SiO4 + 4 SO2
- 3 CaSO4 + CaS → 4 CaO + 4 SO2
- Ca2SiO4 + CaO → Ca3OSiO4
Production and occurrence
The main sources of calcium sulfate are naturally occurring gypsum and anhydrite, which occur at many locations worldwide as evaporites. These may be extracted by open-cast quarrying or by deep mining. World production of natural gypsum is around 127 million tonnes per annum.[17]
In addition to natural sources, calcium sulfate is produced as a by-product in a number of processes:
- In flue-gas desulfurization, exhaust gases from fossil-fuel power stations and other processes (e.g. cement manufacture) are scrubbed to reduce their sulfur dioxide content, by injecting finely ground limestone:[18]
- SO2 + 0.5 O2 + CaCO3 → CaSO4 + CO2
Related sulfur-trapping methods use lime and some produces an impure calcium sulfite, which oxidizes on storage to calcium sulfate.
- In the production of phosphoric acid from phosphate rock, calcium phosphate is treated with sulfuric acid and calcium sulfate precipitates. The product, called phosphogypsum is often contaminated with impurities making its use uneconomic.
- In the production of hydrogen fluoride, calcium fluoride is treated with sulfuric acid, precipitating calcium sulfate.
- In the refining of zinc, solutions of zinc sulfate are treated with hydrated lime to co-precipitate heavy metals such as barium.
- Calcium sulfate can also be recovered and re-used from scrap drywall at construction sites.
These precipitation processes tend to concentrate radioactive elements in the calcium sulfate product. This issue is particular with the phosphate by-product, since phosphate ores naturally contain uranium and its decay products such as radium-226, lead-210 and polonium-210. Extraction of uranium from phosphorus ores can be economical on its own depending on prices on the uranium market or the separation of uranium can be mandated by environmental legislation and its sale is used to recover part of the cost of the process.[19][20][21]
Calcium sulfate is also a common component of fouling deposits in industrial heat exchangers, because its solubility decreases with increasing temperature (see the specific section on the retrograde solubility).
Solubility

The solubility of calcium sulfate decreases as temperature increases. This behaviour ("retrograde solubility") is uncommon: dissolution of most of the salts is endothermic and their solubility increases with temperature.The retrograde solubility of calcium sulfate is also responsible for its precipitation in the hottest zone of heating systems and for its contribution to the formation of scale in boilers along with the precipitation of calcium carbonate whose solubility also decreases when CO2 degasses from hot water or can escape out of the system.
See also
- Calcium sulfate (data page)
- Alabaster
- Anhydrite
- Bathybius haeckelii
- Chalk (calcium carbonate)
- Gypsum
- Gypsum plaster
- Phosphogypsum
- Selenite (mineral)
- Flue-gas desulfurization
References
- ^ Lebedev, A. L.; Kosorukov, V. L. (2017). "Gypsum Solubility in Water at 25°C" (PDF). Geochemistry International. 55 (2): 171–177. doi:10.1134/S0016702917010062. S2CID 132916752.
- ^ D.R. Linde (ed.) "CRC Handbook of Chemistry and Physics", 83rd Edition, CRC Press, 2002
- ^ a b Zumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Company. p. A21. ISBN 978-0-618-94690-7.
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0095". National Institute for Occupational Safety and Health (NIOSH).
- ^ Franz Wirsching "Calcium Sulfate" in Ullmann's Encyclopedia of Industrial Chemistry, 2012 Wiley-VCH, Weinheim. doi:10.1002/14356007.a04_555
- ^ Morikawa, H.; Minato, I.; Tomita, T.; Iwai, S. (1975). "Anhydrite: A refinement". Acta Crystallographica Section B. 31 (8): 2164. doi:10.1107/S0567740875007145.
- ^ Cole, W.F.; Lancucki, C.J. (1974). "A refinement of the crystal structure of gypsum CaSO
4·2H
2O". Acta Crystallographica Section B. 30 (4): 921. doi:10.1107/S0567740874004055. - ^ a b Taylor H.F.W. (1990) Cement Chemistry. Academic Press, ISBN 0-12-683900-X, pp. 186-187.
- ^ "About tofu coagulant". www.soymilkmaker.com. Sanlinx Inc. 31 August 2015. Archived from the original on 14 March 2015. Retrieved 10 January 2008.
- ^ a b "Compound Summary for CID 24497 - Calcium Sulfate". PubChem.
- ^ Titus, Harry W.; McNally, Edmund; Hilberg, Frank C. (1933-01-01). "Effect of Calcium Carbonate and Calcium Sulphate on Bone Development". Poultry Science. 12 (1): 5–8. doi:10.3382/ps.0120005. ISSN 0032-5791.
- ^ Thomas, Mark V.; Puleo, David A.; Al-Sabbagh, Mohanad (2005). "Calcium sulfate: a review". Journal of Long-Term Effects of Medical Implants. 15 (6): 599–607. doi:10.1615/jlongtermeffmedimplants.v15.i6.30. ISSN 1050-6934. PMID 16393128.
- ^ "Biphasic Calcium Sulfate - Overview". Augma Biomaterials. 2020-03-25. Retrieved 2020-07-16.
- ^ Whitehaven Cement Plant
- ^ Anhydrite Process
- ^ COMMONWEALTH OF AUSTRALIA. DEPARTMENT OF SUPPLY AND SHIPPING. BUREAU OF MINERAL RESOURCES GEOLOGY AND GEOPHYSICS. REPORT NO.1949/44 (Geol. Ser. No. 27) by E.K. Sturmfels THE PRODUCTION OF SULPHURIC ACID AND PORTLAND CEMENT FROM CALCIUM SULPHATE AND ALUMINIUM SILICATES
- ^ Gypsum, USGS, 2008
- ^ Speight, James G. (2000). "Fuels, Synthetic, Gaseous Fuels". Kirk‐Othmer Encyclopedia of Chemical Technology. doi:10.1002/0471238961.0701190519160509.a01. ISBN 9780471484943.
- ^ Wang, R. D.; Field, L. A.; Gillet d'Auriac, F. S. "Recovery of uranium from phosphate rocks". OSTI 6654998.
- ^ "Uranium from Phosphates | Phosphorite Uranium - World Nuclear Association".
- ^ "Brazil plans uranium-phosphate extraction plant in Santa Quitéria : Uranium & Fuel - World Nuclear News".

