Search results
Appearance
There is a page named "Technetium(VI) fluoride" on Wikipedia
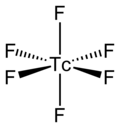
- Technetium hexafluoride or technetium(VI) fluoride (TcF6) is a yellow inorganic compound with a low melting point. It was first identified in 1961. In...8 KB (806 words) - 07:36, 19 May 2024
- Technetium pentafluoride is a binary inorganic chemical compound of technetium metal and fluorine with the chemical formula TcF 5. The compound can be...4 KB (273 words) - 18:05, 11 May 2024
- Technetium(IV) chloride is the inorganic compound with the formula TcCl4. It was discovered in 1957 as the first binary halide of technetium. It is the...5 KB (342 words) - 18:06, 29 December 2023
- Pertechnetyl fluoride is an inorganic compound, a salt of technetium and hydrofluoric acid with the chemical formula TcO 3F. The compound was originally...4 KB (311 words) - 18:03, 23 September 2023
- TcBr4 technetium(IV) bromide 74078–05–0 TcCl4 technetium(IV) chloride 14215–13–5 TcCl6 technetium(VI) chloride 31227–43–7 TcF5 technetium(V) fluoride 31052–14–9...139 KB (120 words) - 17:07, 15 July 2024
- Manganese(II) fluoride is the chemical compound composed of manganese and fluoride with the formula MnF2. It is a light pink solid, the light pink color...5 KB (276 words) - 20:53, 3 January 2024
- vanadate - NdVO4 Neptunium(III) fluoride – NpF3 Neptunium(IV) fluoride – NpF4 Neptunium(IV) oxide – NpO2 Neptunium(VI) fluoride – NpF6 Nickel(II) carbonate...119 KB (8,726 words) - 04:34, 18 April 2024
- 1744372. Supeł, J.; Abram, U.; Hagenbach, A.; Seppelt, K. (2007) "Technetium Fluoride Trioxide, TcO3F, Preparation and Properties." Inorg. Chem., 46(14)...8 KB (805 words) - 18:09, 10 May 2024
- neptunium oxides with anhydrous hydrogen fluoride at various temperatures. Neptunium also forms a wide variety of fluoride compounds with various elements. Some...113 KB (13,646 words) - 18:50, 19 July 2024
- Rhenium tetrafluoride (redirect from Rhenium(IV) fluoride)D. W.; Peacock, R. D. (26 January 2016). The Chemistry of Manganese, Technetium and Rhenium: Pergamon Texts in Inorganic Chemistry. Elsevier. p. 918....3 KB (169 words) - 15:30, 3 January 2024
- Chromium (section Chromium(VI))2 [CrO4]2− + 2 H+ ⇌ [Cr2O7]2− + H2O Chromium(VI) oxyhalides are known also and include chromyl fluoride (CrO2F2) and chromyl chloride (CrO 2Cl 2). However...108 KB (11,748 words) - 19:58, 10 July 2024
- micrograms were not prepared until 1951 by reduction of americium(III) fluoride with barium metal in high vacuum at 1100 °C. The longest-lived and most...76 KB (9,243 words) - 12:57, 19 July 2024
- half full and the electrons are contributing less to metallic bonding. (Technetium, the previous element, has an exceptionally low value that is off the...50 KB (5,744 words) - 02:46, 20 June 2024
- depends quite strongly on the halide counterion: although molybdenum(VI) fluoride is stable, molybdenum does not form a stable hexachloride, pentabromide...73 KB (8,230 words) - 23:15, 18 July 2024
- compound tungsten(VI), WO3. It will, however, react directly with fluorine (F2) at room temperature to form tungsten(VI) fluoride (WF6), a colorless...80 KB (9,050 words) - 04:15, 4 July 2024
- bromine pentafluoride, and either sodium fluoride or nickel fluoride was claimed to produce a higher fluoride as well which hydrolysed to form RnO 3. While...115 KB (13,147 words) - 13:28, 10 July 2024
- Rhenium pentafluoride (category Fluorides)Retrieved 6 April 2023. Colton, Ray (1965). The Chemistry of Rhenium and Technetium. Interscience Publishers. p. 59. ISBN 978-0-470-16650-5. Retrieved 6 April...3 KB (170 words) - 16:10, 29 December 2023
- synthesized in amounts large enough to weigh. Some synthetic elements, like technetium and plutonium, have later been found in nature. Arblaster, John W. (2018)...36 KB (4,059 words) - 13:28, 10 July 2024
- Copernicium (redirect from Copernicium(IV) fluoride)non-existent. Copernicium(II) fluoride, CnF2, should be more unstable than the analogous mercury compound, mercury(II) fluoride (HgF2), and may even decompose...48 KB (8,314 words) - 18:57, 12 July 2024
- elements beyond plutonium on the Periodic table, and three others -- technetium (43 electrons), promethium (61), and neptunium (93) -- are too unstable