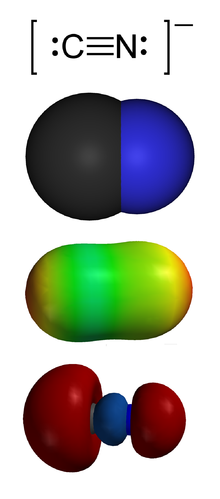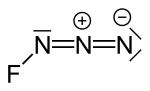Search results
Appearance
There is a page named "N2F2" on Wikipedia
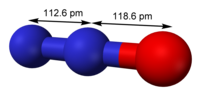
- Dinitrogen difluoride (redirect from N2F2)Dinitrogen difluoride is a chemical compound with the formula N2F2. It is a gas at room temperature, and was first identified in 1952 as the thermal decomposition...7 KB (574 words) - 04:35, 14 February 2024
- Nitrogen trifluoride, NF3 Nitrogen pentafluoride, NF5 Dinitrogen difluoride, N2F2 Tetrafluorohydrazine, N2F4 Fluorine azide, N3F Tetrafluoroammonium, NF4+...656 bytes (73 words) - 18:30, 7 June 2023
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...35 KB (3,877 words) - 13:21, 19 July 2024
- monoxide 10102–43–9 NO2 nitrogen dioxide 10102–44–0 N2 nitrogen 7727-37-9 N2F2 dinitrogen difluoride 13812–43–6 N2F4 dinitrogen tetrafluoride 10036–47–2...139 KB (120 words) - 17:07, 15 July 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...137 KB (14,828 words) - 13:25, 17 July 2024
- azide (FN3) is very explosive and thermally unstable. Dinitrogen difluoride (N2F2) exists as thermally interconvertible cis and trans isomers, and was first...105 KB (12,215 words) - 21:54, 14 June 2024
- explosion at normal temperatures to make dinitrogen difluoride: 2 FN3 → N2F2 + 2 N2. At higher temperatures such as 1000 °C fluorine azide breaks up into...8 KB (724 words) - 12:54, 11 January 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...31 KB (3,494 words) - 14:28, 14 June 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...29 KB (2,569 words) - 19:03, 11 July 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...35 KB (3,695 words) - 12:37, 15 July 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...46 KB (5,185 words) - 14:24, 10 July 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...29 KB (2,758 words) - 02:52, 22 June 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...28 KB (2,895 words) - 03:34, 15 July 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...9 KB (1,002 words) - 02:37, 7 June 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...8 KB (698 words) - 11:59, 8 June 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...4 KB (309 words) - 18:26, 18 May 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...28 KB (2,640 words) - 11:39, 23 May 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...96 KB (10,063 words) - 22:43, 24 June 2024
- N2O2− 3 Halides NF NF2 NF3 NF5 (?) NCl3 NBr3 NI3 FN3 ClN3 BrN3 IN3 NH2F N2F2 NH2Cl NHF2 NHCl2 NHBr2 NHI2 Oxidation states −3, −2, −1, 0, +1, +2, +3, +4...46 KB (4,591 words) - 13:51, 1 July 2024
- difluoride N2F2 −111.45 −172 66 13776-62-0 Ozone O3 −111.35 −193 48 10028-15-6 Xenon Xe −108.099 −111.75 131 7440-63-3 cis-Dinitrogen difluoride N2F2 −105.75...95 KB (4,549 words) - 23:21, 8 July 2024
- name for each of the following molecular compounds: e. IF f. PCl3 g. I2 h. N2F2 Solutions: a. ClF b. H2S c. CBr4 d. Br2 e. Iodine monofluoride f. Phosphorus