Search results
Appearance
There is a page named "Letts nitrile synthesis" on Wikipedia
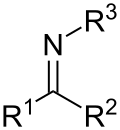
- The Letts nitrile synthesis is a chemical reaction of aromatic carboxylic acids with metal thiocyanates to form nitriles. The reaction includes the loss...8 KB (877 words) - 06:09, 4 September 2023
- (Letts nitrile synthesis) Nitrile groups in organic compounds can undergo a variety of reactions depending on the reactants or conditions. A nitrile group...29 KB (3,472 words) - 15:17, 26 June 2024
- Look up Letts, letts, or lett in Wiktionary, the free dictionary. Letts may refer to: Judge Letts, multiple persons Arthur Letts, American developer of...2 KB (248 words) - 01:56, 21 April 2024
- Acetonitrile (redirect from Ethyl nitrile)H3C−C≡N. This colourless liquid is the simplest organic nitrile (hydrogen cyanide is a simpler nitrile, but the cyanide anion is not classed as organic). It...21 KB (1,804 words) - 01:49, 6 May 2024
- Larock indole synthesis Lawesson's reagent Lebedev process Lehmstedt–Tanasescu reaction Leimgruber–Batcho indole synthesis Letts nitrile synthesis Leuckart...38 KB (3,429 words) - 20:03, 6 June 2024
- Albert Letts FRSE FCS FIC (27 August 1852 – 19 February 1918) was a 19th-century English chemist. He was a pioneer of analytical chemistry. The Letts Nitrile...3 KB (364 words) - 22:18, 18 February 2024
- Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of...39 KB (4,087 words) - 03:50, 29 February 2024
- Nitrile anions is jargon from the organic product resulting from the deprotonation of alkylnitriles. The proton(s) α to the nitrile group are sufficiently...8 KB (999 words) - 18:05, 4 April 2024
- Imine (section From nitriles)synthesized via a Grignard reaction with a nitrile. This method is known as Moureu-Mignonac ketimine synthesis. For example, benzophenone imine can also...23 KB (2,633 words) - 07:34, 30 May 2024
- Wittig reaction (redirect from Wittig synthesis)epoxides, and sometimes esters and amides. Even ketone, aldehyde, and nitrile groups can be present if conjugated with the ylide — these are the stabilised...15 KB (1,600 words) - 17:50, 27 May 2024
- carboxylate ester and a nitrile. It is a colourless liquid with a pleasant odor. This material is useful as a starting material for synthesis due to its variety...9 KB (990 words) - 18:06, 30 July 2024
- Tetrahedron Lett. (in German), vol. 43, no. 25, pp. 4481–4485, doi:10.1016/S0040-4039(02)00831-6 L. Ji; Y.-N. Wang; C. Qian; X.-Z. Chen (2013), "Nitrile-promoted...15 KB (1,405 words) - 09:56, 22 June 2024
- 1992, Robinson and co-workers developed a similar pyridine synthesis using enamino nitriles as one of the three-carbon fragments in place of an α-pyridinium...14 KB (1,765 words) - 19:29, 23 July 2024
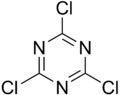
- Cyanuric chloride (section Organic synthesis)Cyanuric Chloride, a Mild Dehydrating Agent in the Preparation of Nitriles from Amides". Synthesis. 8: 657–658. doi:10.1055/s-1980-29160. Probst, D. A.; Hanson...9 KB (680 words) - 17:52, 31 July 2024
- incorporating heterogeneous catalysts are listed below. Reduction of nitriles in the synthesis of phenethylamine with Raney nickel catalyst and hydrogen in ammonia:...30 KB (3,239 words) - 20:28, 22 July 2024
- is the reaction of a ketone with TosMIC leading to the formation of a nitrile. It was first described in 1977 by Van Leusen and co-workers. When aldehydes...4 KB (267 words) - 05:42, 24 September 2020
- potentially provided a source of amino acid derivatives from simple aldehyde and nitrile feedstocks. Alexander Butlerov showed in 1861 that the formose reaction...203 KB (21,024 words) - 08:40, 12 August 2024
- the conversion of aldehydes into nitriles and alicyclic ketones into lactams (cyclic amides), and for the synthesis of variety of nitrogen-containing...17 KB (1,606 words) - 04:27, 22 June 2024
- organic chemistry, hydrocyanation is a process for conversion of alkenes to nitriles. The reaction involves the addition of hydrogen cyanide and requires a...10 KB (1,082 words) - 12:04, 3 June 2024
- and it is compatible with ethers and even ester groups. Even C=O and nitrile groups can be present if conjugated with the ylide- these are the stabilised