Formoterol
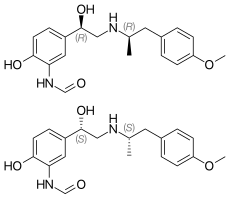 | |
 Formoterol (top), (R,R)-(−)-formoterol (center) and (S,S)-(+)-formoterol (bottom) | |
| Clinical data | |
|---|---|
| Trade names | Oxeze, Foradil, Symbicort, others |
| AHFS/Drugs.com | Monograph |
| License data | |
| Pregnancy category |
|
| Routes of administration | Inhalation (capsules for oral inhalation, DPI, MDI) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 61% to 64% |
| Metabolism | Liver demethylation and glucuronidation (CYP2D6, CYP2C19, CYP2C9 and CYP2A6 involved) |
| Elimination half-life | 10 h |
| Excretion | Kidney and fecal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.131.654 |
| Chemical and physical data | |
| Formula | C19H24N2O4 |
| Molar mass | 344.411 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Formoterol, also known as eformoterol, is a long-acting β2 agonist (LABA) used as a bronchodilator in the management of asthma and chronic obstructive pulmonary disease (COPD). Formoterol has an extended duration of action (up to 12 h) compared to short-acting β2 agonists such as salbutamol (albuterol), which are effective for 4 h to 6 h. Formoterol has a relatively rapid onset of action compared to other LABAs, and is effective within 2-3 minutes.[2] The 2022 Global Initiative for Asthma report [3] recommends a combination formoterol/inhaled corticosteroid inhaler as both a preventer and reliever treatment for asthma in adults. In children, a short-acting β2 adrenergic agonist (e.g., salbutamol) is still recommended.
It was patented in 1972 and came into medical use in 1998.[4] It is available as a generic medication.[5] It is also marketed in the combination formulations budesonide/formoterol and mometasone/formoterol.
Side effects
In November 2005, the US Food and Drug Administration (FDA) released a health advisory alerting the public to findings that show the use of long-acting β2 agonists could lead to a worsening of wheezing symptoms in some patients.[6]
Nowadays, available long-acting β2 agonists include salmeterol, formoterol, bambuterol, and sustained-release oral salbutamol.
Combinations of inhaled steroids and long-acting bronchodilators are becoming more widespread – combination preparations include fluticasone/salmeterol and budesonide/formoterol.
Mechanism of action
Inhaled formoterol works like other β2 agonists, causing bronchodilation by relaxing the smooth muscle in the airway so as to treat the exacerbation of asthma.
Society and culture
Brand names

Formoterol is marketed in three forms: a dry-powder inhaler (DPI), a metered-dose inhaler (MDI) and an inhalation solution, under various brand names including Atock, Atimos/Atimos Modulite, Foradil/Foradile, Fostair, Oxeze/Oxis, Perforomist and Symbicort.
- Foradil/Foradile capsules for oral inhalation (Schering-Plough in the U.S., Novartis rest of world)
- Oxeze/Oxis Turbuhaler dry-powder inhaler (DPI) (AstraZeneca)
- Atock (Astellas)
- Atimos/Atimos Modulite metered-dose inhaler (MDI) (Chiesi)
- Perforomist inhalation solution (Mylan N.V.)
- Symbicort Turbuhaler dry-powder inhaler (DPI) (AstraZeneca)
In some countries, Perforomist is marketed by Viatris after Upjohn merged with Mylan to create Viatris.[7][8]
Uses and combinations
- Arformoterol ((R,R)-(−)-formoterol) — an enantiopure compound used in the management of COPD
- Combination drugs:
References
- ^ "List of nationally authorised medicinal products" (PDF). ema.europa.eu. Retrieved 17 June 2023.
- ^ Anderson GP (1993). "Formoterol: pharmacology, molecular basis of agonism, and mechanism of long duration of a highly potent and selective beta 2-adrenoceptor agonist bronchodilator". Life Sci. 52 (26): 2145–60. doi:10.1016/0024-3205(93)90729-m. PMID 8099696.
- ^ Global Initiative for Asthma (2022). Global Strategy for Asthma Prevention and Management (Updated 2022) (PDF) (Report).
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 543. ISBN 9783527607495.
- ^ "Competitive Generic Therapy Approvals". U.S. Food and Drug Administration (FDA). 29 June 2023. Archived from the original on 29 June 2023. Retrieved 29 June 2023.
- ^ "Advair Diskus, Advair HFA, Brovana, Foradil, Perforomist, Serevent Diskus, and Symbicort Information (Long Acting Beta Agonists)". U.S. Food and Drug Administration (FDA). Archived from the original on 1 November 2017. Retrieved 16 December 2019.
- ^ "Pfizer Completes Transaction to Combine Its Upjohn Business with Mylan". Pfizer. 16 November 2020. Retrieved 17 June 2024 – via Business Wire.
- ^ "Brands". Viatris. 16 November 2020. Retrieved 17 June 2024.
